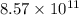Two solutions are combined in a beaker. One solution contains 500.0 g of potassium phosphate and the other contains 500.0 g of calcium nitrate. A double-displacement reaction occurs. What mass of each of the following substances is present when the reaction stops?
Solved
Show answers
More tips
- G Goods and services Kogda zhdatt Iphone 5? The Latest News and Rumors...
- F Family and Home Parquet or laminate, which is better?...
- L Leisure and Entertainment How to Properly Wind Fishing Line onto a Reel?...
- L Leisure and Entertainment How to Make a Paper Boat in Simple Steps...
- T Travel and tourism Maldives Adventures: What is the Best Season to Visit the Luxurious Beaches?...
- H Health and Medicine Kinesiology: What is it and How Does it Work?...
- O Other How to Choose the Best Answer to Your Question on The Grand Question ?...
- L Leisure and Entertainment History of International Women s Day: When Did the Celebration of March 8th Begin?...
- S Style and Beauty Intimate Haircut: The Reasons, Popularity, and Risks...
- A Art and Culture When Will Eurovision 2011 Take Place?...
Answers on questions: Chemistry
- M Mathematics Si una bolsa de plástico pesa cuatro kilogramos y contiene tres latas con atún y dos de mermelada, y en otra bolsa contiene dos de atún y cinco de mermelada con un peso de 5.5 kilogramos....
- M Mathematics Salma gives a cashier $20 bill. She is buying an item for $9.85. How much money should salma get back...
- M Mathematics If 8 unites represent 136 miles, find the value of 11 units...
- M Mathematics Tennis balls are sold in tubes that hold 3 tennis balls each. a store stacks 2 rows of tennis ball tubes on its shelf. each row has 7 tubes in it. how many tennis balls are there...
- B Biology 31. What are the chances that a puppy will have a spotted coat, if his mother is solid and his father is heterozygous for a spotted coat?...

Ответ:
The question is incomplete. The complete question is
Two solutions are combined in a beaker. One solution contains 500.0 g of potassium phosphate and the other contains 500.0 g of calcium nitrate. A double displacement reaction occurs. What mass of each of the following substances is present when the reaction stops. A) potassium phosphate remaining B) calcium nitrate g remaining C) calcium phosphate formed D) potassium nitrate g formed
a)84.91g
b)8.20g
c)316.4g
d)616.73g
Explanation:
The equation of the reaction:
2K3PO4(aq) + 3Ca(NO3)2 (aq)> 6KNO3(aq) + Ca3(PO4)2(s)
Molar mass of potassium phosphate= 212.27 g/mol
Amount of potassium phosphate= 500/212.27= 2.4 moles
Molar mass of calcium nitrate= 164.088 g/mol
Amount of calcium nitrate= 500/164.088=3.05moles
a) amount of potassium phosphate reacted according to reaction equation= 2 moles
Amount of potassium phosphate remaining= 2.4-2=0.4 moles
Mass of potassium phosphate remaining= 0.4×212.27=84.91g
b) Amount of calcium nitrate reacted according to reaction equation=3
Amount of calcium nitrate remaining=3.05-3= 0.05
Mass of calcium nitrate remaining= 0.05×164.088= 8.20g
c) since calcium nitrate is the limiting reactant, we use to estimate the mass of products formed.
From the reaction equation,
3 moles of calcium nitrate yields 1 mole of calcium phosphate
3.05 moles of calcium nitrate yields 3.05/3 = 1.02 moles of calcium phosphate
Molar Mass of calcium phosphate= 310.18 g/mol
Mass of calcium phosphate produced= 1.02×310.18= 316.4g
d)
3 moles of calcium nitrate yields 6 moles of potassium nitrate
3.05 moles of calcium nitrate yields 3.05×6/3= 6.1 moles of potassium nitrate
Molar mass of potassium nitrate = 101.1032 g/mol
Mass of potassium nitrate formed= 6.1× 101.1032= 616.73g
Ответ:
The number of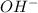 ions dissociated are
ions dissociated are 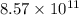
Explanation:
We are given:
pH = 2.07
Calculating the value of pOH by using equation, we get:
To calculate hydroxide ion concentration, we use the equation to calculate pOH of the solution, which is:
We are given:
pOH = 11.93
Putting values in above equation, we get:
To calculate the number of moles for given molarity, we use the equation:
Molarity of solution =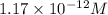
Volume of solution = 1243 mL = 1.243 L (Conversion factor: 1 L = 1000 mL)
Putting values in above equation, we get:
According to mole concept:
1 mole of a compound contains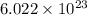 number of particles
number of particles
So,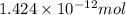 number of
number of 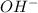 will contain =
will contain = 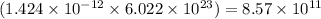 number of ions
number of ions
Hence, the number of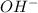 ions dissociated are
ions dissociated are