Use the reaction given below to solve the problem that follows: Calculate the mass in grams of aluminum oxide produced by the reaction of 15.0 g of aluminum metal.
[ ]grams Al2O3
4 Al + 3 O2 --> 2 Al2O3
**Your answer should be written as XX.X
Solved
Show answers
More tips
- S Society and Politics What Would Brezhnev Do If He Were Alive Today?...
- S Style and Beauty Choosing a Hair Straightener: Specific Criteria to Consider...
- F Family and Home How to Remove Tar Stains: Tips and Recommendations from Experts...
- F Family and Home How to Remove Fading from Clothes: Tips and Tricks...
- S Sport How to Do a Jumping Split...
- H Health and Medicine How Did Inna Lose Weight on Dom 2?...
- F Family and Home How to Properly Fold Napkins in a Napkin Holder?...
- F Food and Cooking How to Set Up Ventrilo - The Ultimate Guide...
- S Science and Technology How to Make a Homemade Smoker: The Ultimate Guide...
- A Auto and Moto Battle for the Relocation of The Cherkizovsky Market: Who Won?...
Answers on questions: Chemistry
- C Chemistry Calculating equilibrium concentrations when the net reaction proceeds in reverse concentration (m) [xy] [x] [y] initial: 0.200 0.300 0.300 change: +x -x -x equilibrium: 0.200+x...
- C Chemistry Which of the following choices must occur during all chemical changes...
- C Chemistry What does it mean when a wave s amplitude increases?...
- M Mathematics The midpoint of the coordinates (4, 10) and (13,3) is __. O A. (9,7) B. (8.5, 6.5) C. (17, 13) O D. none of the above...
- M Mathematics F(x) = x-2. Find the inverse of f(x)....
- M Mathematics 3. How many solutions does this system have? (1 point) yx+ 5 y = -5x - 1 one none Infinite two...
- M Mathematics Solve the equation by factoring. 5z2 − 21z + 4 = 0 A.) z = 1 or z = 4 B.) z = or z = –4 C.) z = 1 or z = –4 D.) z = or z = 4 Use format, Explanation: Contact me on Discord if...
- C Chemistry Out of hexafluoridocobaltate(iii) and tris(ethane-1,2-diamine)cobalt(iii) ions, which one is a) paramagnetic b) more stable c)inner orbital complex d) high spin complex?....
- H History Which were issues or events that led to civil war in England? Choose all answers that are correct. Puritans opposed Catholicism and feared that Charles I s Catholic wife would...
- M Mathematics Does anyone know the answer...

Ответ:
28.4 g of aluminum oxide is produced by the reaction of 15.0 g of aluminum metal
Explanation:
To calculate the moles :
The balanced chemical equuation is:
According to stoichiometry :
4 moles of produce == 2 moles of
produce == 2 moles of 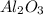
Thus 0.556 moles of will produce=
will produce=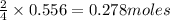 of
of 
Mass of
Thus 28.4 g of aluminum oxide is produced by the reaction of 15.0 g of aluminum metal.
Ответ:
287.68g
Explanation:
We'll begin by calculating the number of mole of FeCl2 in 1.24L of a 2M FeCl2 solution. This is illustrated below:
Molarity = 2M
Volume = 1.24L
Mole of FeCl2 =..?
Mole = Molarity x Volume
Mole of FeCl2 = 2 x 1.24
Mole of FeCl2 = 2.48 moles
Next, the balanced equation for the reaction.
FeCl2(aq) + Na2CO3(s) → FeCO3(s) + 2NaCl(aq).
Next, we shall determine the mole of FeCO3 produced from the reaction.
This is illustrated below:
From the balanced equation above,
1 mole of FeCl2 reacted to produce 1 mole of FeCO3.
Therefore, 2.48 moles of FeCl2 will also react to produce 2.48 moles of FeCO3.
Finally, we shall convert 2.48 moles of FeCO3 to grams.
Number of mole of FeCO3 = 2.48 moles
Molar mass of FeCO3 = 56 + 12 + (16x3) = 116g/mol.
Mass of FeCO3 =..?
Mass = mole x molar mass
Mass of FeCO3 = 2.48 x 116
Mass of FeCO3 = 287.68g
Therefore, 287.68g of FeCO3 were from the reaction.