What is the chemical formula for the copper?
Solved
Show answers
More tips
- H Horoscopes, Magic, Divination How to Cast a Love Spell on a Guy? Guide for Guys...
- F Family and Home How to Sew Curtain Tapes: Best Tips from Professionals...
- S Style and Beauty How are artificial nails removed?...
- S Style and Beauty How to Make Your Lips Fuller? Ideas and Tips for Beautiful Lips...
- F Food and Cooking The Disease That Haunted Abraham Lincoln...
- C Computers and Internet How to Get Rid of Windows Genuine Check?...
- H Health and Medicine How to perform artificial respiration?...
- S Style and Beauty Tricks and Tips: How to Get Rid of Freckles...
- C Computers and Internet How Do You Refill Cartridges?...
- A Auto and Moto Battle for the Relocation of The Cherkizovsky Market: Who Won?...
Answers on questions: Chemistry
- C Chemistry Why do you use 6.02*10^23 here? ? #...
- C Chemistry In which two types of reactions do opposite chemical changes occur? a) double replacement and combustion b) single replacement and double replacement c) synthesis and decomposition...
- C Chemistry How many steps to complete mitosis and meiosis? ?...
- C Chemistry What type of bonds form salt crystals...
- C Chemistry Where might life exist in the solar system beyond earth...
- C Chemistry in which of the following reactions would you expect the orientation factor to be least important in leading to reaction: NO + O yield NO2 or H + Cl yield HCl?...
- C Chemistry When performing a qualitative experiment, you need to A. measure the exact mass of a chemical. B. measure the exact volume of a chemical. C. measure the exact size of a...
- C Chemistry What is the range of output potential difference? 7.5 la 12.5 12.5...
- C Chemistry Can someone actually help me and answer the 4 questions...
- C Chemistry The mass percent of water in hydrate of MnCl2 is 36.41%.what is the empirical formula of the hydrate...

Ответ:
Hey mate
Explanation:
This is ur answer
CuHope it helps!
Brainliest pls!
Follow me! ;)
Ответ:
cu I believe but don't quote me on that
Ответ:
Explanation:
Molarity is defined as the number of moles per liter of solution.
Mathematically, Molarity =
Since it is given that the molarity of a solution of 14.0 g and volume is 150 mL or 0.15 L.
Whereas number of moles =
So, molar mass of is 97.94 g/mol.
is 97.94 g/mol.
Thus, number of moles =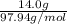
= 0.142 mol
Therefore, calculate the molarity as follows.
Molarity =
=
= 0.946 mol/L
Hence, we can conclude that molarity of the solution is 0.946 mol/L.