What is the hybridization of bromine in bro2-?
Solved
Show answers
More tips
- F Food and Cooking The Health Benefits of Flaxseed oil...
- S Style and Beauty Why is Sugaring Better than Waxing for Hair Removal?...
- W Work and Career Where can you learn to be a flight attendant?...
- G Goods and services How to Properly Calculate the Power of Your Air Conditioner?...
- F Food and Cooking Effective Methods to Organize Videos in your iPad According to Content...
- F Family and Home Parquet or laminate, which is better?...
- L Leisure and Entertainment How to Properly Wind Fishing Line onto a Reel?...
- L Leisure and Entertainment How to Make a Paper Boat in Simple Steps...
- T Travel and tourism Maldives Adventures: What is the Best Season to Visit the Luxurious Beaches?...
- H Health and Medicine Kinesiology: What is it and How Does it Work?...
Answers on questions: Chemistry
- S Spanish The present progressive tense is used to say what the subject... A. is doing B. has done C. wants to do. D. will do....
- H History Who negotiate an alliance between the patriots and france in 1778? a)charles cornwallis b)marquis de lafayette c)benjamin franklin d)john burgoyne...
- A Arts hey yall. if yes why and if not why?...
- M Mathematics For two independent events A and B, Pr(A or B) = 0.8 and Pr(B) = 0.3. From this, find Pr(A)....

Ответ:
Answer : The hybridization of the bromine in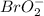 is,
is, 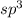
Explanation :
Formula used :
where,
V = number of valence electrons present in central atom
N = number of monovalent atoms bonded to central atom
C = charge of cation
A = charge of anion
Now we have to determine the hybridization of the bromine in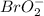 molecule.
molecule.
The given molecule is,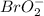
The number of electron pair are 4 that means the hybridization will be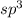 and the electronic geometry of the molecule will be tetrahedral.
and the electronic geometry of the molecule will be tetrahedral.
But as there are 2 atoms around the central oxygen atom, the third and fourth position will be occupied by lone pair of electrons. The repulsion between lone and bond pair of electrons is more and hence the molecular geometry will be bent or angular.
Hence, the hybridization of the bromine in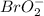 is,
is, 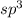
Ответ: