boopiee2349
18.09.2019 •
Chemistry
What is the molarity of a solution of 14.0 g nh4br in enough h2o to make 150 ml of solution?
Solved
Show answers
More tips
- C Computers and Internet 3D Glasses! What is this thing?...
- C Computers and Internet How to insert videos into LiveJournal?...
- C Computers and Internet How Much Does an iPhone Cost in America?...
- F Family and Home How to Choose the Best Diapers for Your Baby?...
- F Family and Home Parquet or laminate, which is better?...
- L Leisure and Entertainment How to Properly Wind Fishing Line onto a Reel?...
- L Leisure and Entertainment How to Make a Paper Boat in Simple Steps...
- T Travel and tourism Maldives Adventures: What is the Best Season to Visit the Luxurious Beaches?...
- H Health and Medicine Kinesiology: What is it and How Does it Work?...
- O Other How to Choose the Best Answer to Your Question on The Grand Question ?...
Answers on questions: Chemistry
- C Chemistry Why are the alkali metals and the halogens among the most reactive elements on the periodic table...
- C Chemistry Atomic mass of an element depends upon what...
- C Chemistry You take a glow stick, crack the capsule inside and shake it until it glows. this is an example of light energy being created from energy....
- C Chemistry Describe the overall trend for each of the five factors that were measured....
- C Chemistry I) Calculate theoretical yield of product if one started with 11g phenol and 12ml Ac2O...
- E Engineering 1. In order to limit or reduce blank, which cause heating in the motor iron, the stator is constructed of many thin sheets of metal....
- M Mathematics Cole s soup recipe is shown in the table above. What is the ratio of water to chicken stock? Express the ratio as a fraction in simplest form....
- P Physics List four conditions that may result in the emission of electrons from a conductor. Compare and contrast characteristics and uses of diodes and triodes. Identify the...
- A Arts Heres Rollo killing the cat (again)...
- B Business PLEASE HELP ME IN MY THEATER CLASS...

Ответ:
Explanation:
Molarity is defined as the number of moles per liter of solution.
Mathematically, Molarity =
Since it is given that the molarity of a solution of 14.0 g and volume is 150 mL or 0.15 L.
Whereas number of moles =
So, molar mass of is 97.94 g/mol.
is 97.94 g/mol.
Thus, number of moles =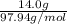
= 0.142 mol
Therefore, calculate the molarity as follows.
Molarity =
=
= 0.946 mol/L
Hence, we can conclude that molarity of the solution is 0.946 mol/L.
Ответ:
Ответ:
If you add soap or detergent in water, the surface tension of water changes, the molecular bond of water becomes weaker. So, when light hits a plain water, it could either be reflected, or the water gets heated and evaporates, and then forms into gas.
Explanation:
Something like this?