calmicaela12s
19.10.2019 •
Chemistry
What is the percent by mass of oxygen in propanal, ch3ch2cho? (1) 10.0% (3) 38.1%
(2) 27.6% (4) 62.1%
Solved
Show answers
More tips
- S Style and Beauty How to knit a hooded cowl?...
- H Horoscopes, Magic, Divination How to Cast a Love Spell on a Guy? Guide for Guys...
- S Style and Beauty Tricks and Tips: How to Get Rid of Freckles...
- H Health and Medicine How to perform artificial respiration?...
- C Computers and Internet How to Get Rid of Windows Genuine Check?...
- F Food and Cooking The Disease That Haunted Abraham Lincoln...
- S Style and Beauty How to Make Your Lips Fuller? Ideas and Tips for Beautiful Lips...
- S Style and Beauty How are artificial nails removed?...
- F Family and Home How to Sew Curtain Tapes: Best Tips from Professionals...
- F Family and Home How to Properly Use a Water Level?...
Answers on questions: Chemistry
- C Chemistry Question: convert 22.63 miles of oxygen to volume very urgent...
- C Chemistry Question: convert 2.45•10^21 formula unit of ammonium sulfate to moles very urgent...
- C Chemistry 1. What is the wavelength of radi- ation with a frequency of 1.50 X 1013 s-l? Does this radiation have a longer or shorter wave- length than red light?...
- C Chemistry Asingle penny has a mass of 2.5 g. abbie and james each measure the mass of a penny multiple times. which statement about these data sets is true? abbie s measurements are both...
- C Chemistry The chemical formula below shows how salt is produced. HCl NaOH mc004-1. Jpg NaCl H2O Which substances are the reactants? HCl and NaCl NaCl and H2O HCl and NaOH NaOH and H2O....
- C Chemistry Phenol, is a weak organic acid. Suppose g of the compound is dissolved in enough water to make mL of solution. The resulting solution is titrated with M .What is the pH of the original...
- C Chemistry I NEED HELP NOW PLEASE IT S COMPLICATED! I NEED TO WRITE A LAB! Table A Student Guide (continued) Starch Glucose Protein Test Tube (+/–) Start Color End Color (+/–) Color (+/–)...
- C Chemistry Metallic iron crystallizes in a cubic lattice. The unit cell edge length is 287 pm. The density of iron is 7.87 g/cm3. How many iron atoms are within a unit cell?...
- C Chemistry Which of the following statements is true? A. A buffer is best used in pH ranges from two units below up to one unit above its pKa. B. A buffer works best in a pH range farthest...
- C Chemistry The table shows the conditions during two trials of a reaction....

Ответ:
Answer : The percent by mass of oxygen in propanal is, 27.58 %
Solution : Given,
Molar mass of carbon = 12 g/mole
Molar mass of hydrogen = 1 g/mole
Molar mass of oxygen = 16 g/mole
First we have to calculate the molar mass of propanal,
Molar mass of propanal, =
= 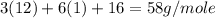
Now we have to calculate the percent by mass of oxygen in propanal.
Therefore, the percent by mass of oxygen in propanal is, 27.58 %
Ответ:
They are Additives