What is the percent yield of aluminum phosphate if a solution containing 33.4 g of sodium phosphate produced 19.6 g of aluminum phosphate when reacted with excess aluminum chloride in solution
Solved
Show answers
More tips
- P Philosophy Unidentified Flying Object - What is the Nature of this Phenomenon?...
- F Family and Home Protect Your Home or Apartment from Pesky Ants...
- O Other What is a Disk Emulsifier and How Does it Work?...
- H Health and Medicine How to Calm Your Nerves? Expert Tips That Actually Work...
- A Animals and plants 5 Tips for Taking Care of Yews to Keep Them Green and Beautiful...
- S Sport How to wrap boxing hand wraps? Everything you need to know!...
- F Food and Cooking 10 Reasons Why You Should Avoid Giving Re-Gifts: An Informative Guide...
- F Family and Home Tender Care for Your Parquet: Is it Possible to Clean Parquet?...
- S Style and Beauty How Are Eyelash Extensions Applied? All Your Questions Answered...
- F Food and Cooking 10 Tips for Proper Sushi Consumption...
Answers on questions: Chemistry
- C Chemistry Ammonium phosphate NH43PO4 is an important ingredient in many fertilizers. It can be made by reacting phosphoric acid H3PO4 with ammonia NH3. What mass of ammonium phosphate...
- C Chemistry What’s is inside a cell? Crossword puzzle ACROSS 2.package materials from the endoplasmic reticulum & send them to other parts of the cell 4.breaks down food & old cell...
- C Chemistry Khí nào không cháy trong khí oxi? CO2,CH4,CO,H2...
- C Chemistry To gravimetrically analyze the silver content of a piece of jewelry made from an alloy of ag and cu, a student dissolves a small, pre-weighed sample in hno3(aq). ag+(aq) and...
- C Chemistry Two tanks (tank a and tank b) of gas are connected by a closed valve. tank a is 5 liters and contains o2 gas at a pressure of 24 atm. tank b is 3 liters and contains n2 gas...
- C Chemistry Consider four beakers labeled a, b, c, and d, each containing an aqueous solution and a solid piece of metal. identity the beakers in which a chemical reaction will occur and...
- S Spanish Which phrase best complete the question? ¿puedes darme para ir a comprar el periódico? a.tarjeta de crédito b.tarjeta de debito c.cheques d.dinero en efectivo...
- M Mathematics Apolygon has an angle sum of 180 and each angle measures 60. what is the polygon?...
- G Geography In the united states where can trenches that form volcanoes be found?...
- S Social Studies Americans began settling the new mexico region as early as 1801....

Ответ:
The percent yield of aluminium phosphate in the reaction is 78.78 %
Explanation:
To calculate the number of moles, we use the equation:
 .....(1)
.....(1)
For sodium phosphate:Given mass of sodium phosphate = 33.4 g
Molar mass of sodium phosphate = 164 g/mol
Putting values in equation 1, we get:
The chemical equation for the reaction of sodium phosphate and aluminium chloride is:
By Stoichiometry of the reaction:
1 mole of sodium phosphate produces 1 mole of aluminium phosphate
So, 0.204 moles of sodium phosphate will produce =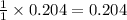 moles of aluminium phosphate
moles of aluminium phosphate
Now, calculating the mass of aluminium phosphate from equation 1, we get:Molar mass of aluminium phosphate = 122 g/mol
Moles of aluminium phosphate = 0.204 moles
Putting values in equation 1, we get:
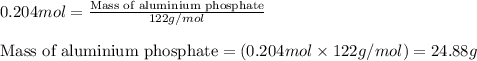
To calculate the percentage yield of aluminium phosphate, we use the equation:Experimental yield of aluminium phosphate = 19.6 g
Theoretical yield of aluminium phosphate = 24.88 g
Putting values in above equation, we get:
Hence, the percent yield of aluminium phosphate in the reaction is 78.78 %
Ответ:
d