MsDiaz9324
20.09.2019 •
Chemistry
What is the ph of a 0.034 m hydrochloric acid solution?
Solved
Show answers
More tips
- F Food and Cooking What Does a Godmother Give?...
- H Health and Medicine Simple and Effective: How to Get Rid of Cracked Heels...
- H Health and Medicine Relieving Swelling in Legs: Causes and Ways to Alleviate the Symptom...
- W Work and Career Мерчендайзинг – все, что нужно знать...
- O Other Everything You Need to Know About Kudyabliks...
- F Food and Cooking How to cook crayfish? Everything you need to know...
- F Food and Cooking Homemade kvass: recipe and brewing process...
- H Health and Medicine How to Choose the Right Tanning Cream?...
- S Style and Beauty Secrets of Tying a Pareo: 5 Ways...
- S Sport Running: How to Do It Right?...
Answers on questions: Chemistry
- M Mathematics If you divide me by 9, the remainder is 4. I am a multiple of 10. What two-digit number am I?...
- E English The excerpt rhyme scheme is...
- M Mathematics Me plz : i’ve waited 30 mins and this is my third time posting this!...
- E English How is the symbolism of the American flag similar to the symbolism of the five arrows that represent the Iroquois Confederacy?...

Ответ:
The pH of the given HCl solution is 1.47
Explanation:
We are given:
Molarity of HCl = 0.034 M
1 mole of HCl produces 1 mole of hydrogen ions and 1 mole of chlorine ions.
pH is defined as negative logarithm of hydrogen ion concentration. It is basically defined as the power of hydrogen ions in a solution.
Mathematically,
We are given:
Putting values in above equation, we get:
Hence, the pH of the given HCl solution is 1.47
Ответ:
Step-by-step explanation:
In circle with center O,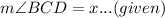
By inscribed angle theorem: