GiselNeedsHelp101
29.01.2020 •
Chemistry
What is the theoretical yield of ammonia (in grams) if 16.55 grams of nitrogen gas and 10.15 grams of hydrogen gas are allowed to react?
Solved
Show answers
More tips
- C Computers and Internet 3D Glasses! What is this thing?...
- C Computers and Internet How to insert videos into LiveJournal?...
- C Computers and Internet How Much Does an iPhone Cost in America?...
- F Family and Home How to Choose the Best Diapers for Your Baby?...
- F Family and Home Parquet or laminate, which is better?...
- L Leisure and Entertainment How to Properly Wind Fishing Line onto a Reel?...
- L Leisure and Entertainment How to Make a Paper Boat in Simple Steps...
- T Travel and tourism Maldives Adventures: What is the Best Season to Visit the Luxurious Beaches?...
- H Health and Medicine Kinesiology: What is it and How Does it Work?...
- O Other How to Choose the Best Answer to Your Question on The Grand Question ?...
Answers on questions: Chemistry
- C Chemistry A 25.00 mL sample of H2SO4 requires 31.43 mL of 0.110 M NaOH to reach the equivalence point. What is the molarity of the H2SO4...
- C Chemistry What’s the chemical equation for all of these...
- C Chemistry What is the minimum mass of ice at 0.0 c that must be added to 1.00 kg of water to cool the water from 28.0 c to 12.0 c? (heat of fusion = 333 j/g; specific heat capacities:...
- C Chemistry Can some one help i will give the instructions please dont delete my question...
- M Mathematics The length of a movie falls on a normal distribution. About 95% of movies fall between 75 minutes and 163 minutes. Required: a. What is the mean value for average movie length...
- B Business The leaders of Greenfiels Inc sometimes alter financial reports and give inaccurate information to the shareholders of the firm:.a) capacity building change initiates cannot...
- M Mathematics 3+3 is what I don t get at all...
- M Mathematics como podría hacer este ejercicio Demuestra que la tasa de variación media de la presión en función de la profundidad en el intervalo de 0 m hasta 15 m, es constante para cualquier...
- B Business A simple trust has ordinary income of $56,000, a long-term capital gain of $20,000 (allocable to corpus), and a trustee commission expense of $5,500 (payable from corpus)....
- C Computers and Technology In _cell reference if you move the formula to another cell,the cells referred to in the formula will not change option 1conditional formatting 2mixed 3 relative 4 absolute...

Ответ:
The theoretical yield of ammonia is 20.13 grams
Explanation:
Step 1: Data given
Mass of N2= 16.55 grams
Mass of H2 = 10.15 grams
Molar mass of N2 = 28 g/mol
Molar mass of H2 = 2.02 g/mol
Step 2: The balanced equation
N2 + 3H2 → 2NH3
Step 3: Calculate moles N2
Moles N2 = mass N2 / molar mass N2
Moles N2 = 16.55 grams / 28.0 g/mol
MolesN2 = 0.591 moles
Step 4: Calculate moles H2
Moles H2 = 10.15 grams / 2.02 g/mol
Moles H2 = 5.02 moles
Step 5: Calculate limiting reactant
For 1 mol N2 we need 3 moles H2 to produce 2 moles of NH3
N2 is the limiting reactant. It will completely be consumed (0.591 moles).
H2 is in excess. There will reat 3*0.591 = 1.773 moles
There will remain 5.02 - 1.773 = 3.247 moles H2
Step 6: Calculate moles of NH3
For 1 mol N2 we need 3 moles H2 to produce 2 moles of NH3
For 0.591 moles N2 we'll have 2-0.591 = 1.182 moles NH3
Step 7: Calculate mass of NH3
Mass NH3 = moles NH3 * molar mass NH3
Mass NH3 = 1.182 * 17.03 g/mol
Mass NH3 = 20.13 grams
The theoretical yield of ammonia is 20.13 grams
Ответ:
Answer : 20.1 grams
Explanation:
According to avogadro's law, 1 mole of every substance occupies 22.4 L at STP and contains avogadro's number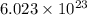 of particles.
of particles.
To calculate the number of moles, we use the equation:
For nitrogen:
For hydrogen gas:
The chemical equation for the reaction is:
By Stoichiometry of the reaction:
1 mole of nitrogen gas reacts with 3 moles of hydrogen.
So, 0.591 moles of nitrogen gas will react with = of hydrogen.
of hydrogen.
As, given amount of hydrogen is more than the required amount. So, it is considered as an excess reagent.
Thus, nitrogen gas is considered as a limiting reagent because it limits the formation of product.
By Stoichiometry of the reaction:
1 mole of nitrogen gas produces 2 moles of ammonia
So, 0.591 moles of nitrogen gas will produce =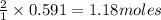 of ammonia
of ammonia
Now, calculating the mass of ammonia t:
Thus theoretical yield of ammonia is 20.1 grams
Ответ: