lebronbangs8930
24.08.2021 •
Chemistry
What mass of water will give up 240 calories when its temperature drops from 12°C.
Solved
Show answers
More tips
- S Science and Technology Why is there no gravity on other planets?...
- F Food and Cooking Deflope: What is it and how does it work?...
- B Business and Finance How to Create a Business Plan? Your Ultimate Guide...
- F Food and Cooking Unusually Delicious Shashlik - Follow the Etiquette of Proper Preparation!...
- C Computers and Internet Make Easy Accessible Screenshots on iPad in Just a Few Minutes...
- T Travel and tourism Lost in the Catacombs: What to Do?...
- F Family and Home Protect Your Home or Apartment from Pesky Ants...
- H Health and Medicine How to Treat Styes: Causes, Symptoms, and Home Remedies...
- L Legal consultation What Documents Are Required for a Russian Passport?...
- F Family and Home How to Properly Use a Water Level?...
Answers on questions: Chemistry
- P Physics Which object provides an inertial frame of reference? which object provides an inertial frame of reference? the tip of the moving second hand of a clock a rock thrown vertically...
- E English ny Form of Government becomes destructive of these ends, it is the Right of the People to alter or to abolish it, and to institute new Government. Which best describes the language...
- P Physics Sources of errors in young mudulus experiment...
- M Mathematics Which logarithmic function is shown in the graph?...
- M Mathematics Find the value of sinA in the figure above. A) 0.7500 B) 0.6000 C) 1.3333 D) 0.8000...

Ответ:
The empirical and molecular formula for the given organic compound is CH and and it is not an alkane.
and it is not an alkane.
Explanation:
The chemical equation for the combustion of hydrocarbon having carbon and hydrogen follows:
where, 'x' and 'y' are the subscripts of Carbon, hydrogen and oxygen respectively.
We are given:
Mass of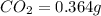
Mass of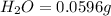
We know that:
Molar mass of carbon dioxide = 44 g/mol
Molar mass of water = 18 g/mol
For calculating the mass of carbon:
In 44 g of carbon dioxide, 12 g of carbon is contained.
So, in 0.364 g of carbon dioxide, of carbon will be contained.
of carbon will be contained.
For calculating the mass of hydrogen:
In 18 g of water, 2 g of hydrogen is contained.
So, in 0.0596 g of water,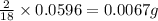 of hydrogen will be contained.
of hydrogen will be contained.
To formulate the empirical formula, we need to follow some steps:
Step 1: Converting the given masses into moles.Moles of Carbon =
Moles of Hydrogen =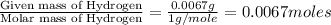 ]
]
Step 2: Calculating the mole ratio of the given elements.For the mole ratio, we divide each value of the moles by the smallest number of moles calculated which is 0.0067 moles.
For Carbon =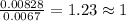
For Hydrogen =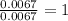
Step 3: Taking the mole ratio as their subscripts.The ratio of C : H = 1 : 1
The empirical formula for the given compound is CH
For determining the molecular formula, we need to determine the valency which is multiplied by each element to get the molecular formula.
The equation used to calculate the valency is:
We are given:
Mass of molecular formula = 128.2 g/mol
Mass of empirical formula = 13 g/mol
Putting values in above equation, we get:
Multiplying this valency by the subscript of every element of empirical formula, we get:
The general formula of an alkane is , where n = any natural number
, where n = any natural number
Here, n = 10 and it does not satisfy being an alkane
Hence, the empirical and molecular formula for the given organic compound is CH and and it is not an alkane.
and it is not an alkane.