unicorns110504
15.12.2020 •
Chemistry
What remains of edith finch

Solved
Show answers
More tips
- L Leisure and Entertainment Learn How to Knit with Needles: A Simple Guide for Beginners...
- C Computers and Internet Best Antivirus: How to Choose Protection for Your Computer?...
- L Legal consultation What Documents Are Required for a Russian Passport?...
- S Sport How to Choose Tennis Rackets?...
- H Health and Medicine AKDS Vaccination: Ensure Your Child s Safety...
- H Health and Medicine Naskol ko Opasen Ukus Kleshcha i Kak Ego Raspoznat...
- C Computers and Internet How to Delete Your Account on Odnoklassniki...
- H Health and Medicine What to Do When Your Jaw Locks Up?...
- G Goods and services What Are the Most Popular Services?...
Answers on questions: Chemistry
- M Mathematics Find the standard deviation of 234.3,225.5,229.4,235.5,239.4,240.4,246.3,249.7,243.1,261.7...
- B Biology What molecule is represented by the molecular model shown below? O Starch O Adenosine triphosphate (ATP) O Glucose O Adenosine diphosphate (ADP)...
- H History What was McClellans mistake at the beginning of the civil war...
- B Business What should you do if you suspect your boss of unethical business practices?...

Ответ:
what is this
Ответ:
The diagram made by the student is correct
Explanation: Endothermic reactions are defined as the reactions in which energy of the product is greater than the energy of the reactants. The total energy is absorbed in the form of heat and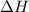 which is difference between energy of products and energy of reactants come out to be positive.
which is difference between energy of products and energy of reactants come out to be positive.
Exothermic reactions are defined as the reactions in which energy of the product is lesser than the energy of the reactants. The total energy is released in the form of heat and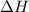 which is difference between energy of products and energy of reactants comes out to be negative.
which is difference between energy of products and energy of reactants comes out to be negative.
Labeling of the parts in the diagram:
X represents the potential energy of the reactants.
Y represents the potential energy of the products.
The energy diagram for an exothermic reaction should have products at a lower energy than the reactants as heat is released.