What would be the volume in liters of an ideal gas, if a 0.111 mole sample of the gas had a temperature of 1130 degrees celsius at a pressure of 5.45 atmospheres? (the ideal gas constant is 0.0821 l•atm/mol•k.)
0.43 liters
1.43 liters
1.89 liters
2.35 liters
Solved
Show answers
More tips
- F Food and Cooking Kick Your Taste Buds Into Gear with This Authentic and Delicious Recipe for Solyanka...
- O Other What happens if you get scared half to death twice?...
- F Family and Home What s That Noise When a Kettle Boils? The Science of Water and Steam...
- L Leisure and Entertainment History of International Women s Day: When Did the Celebration of March 8th Begin?...
- W Work and Career How much does an honest traffic police officer earn in a day?...
- F Food and Cooking Red Caviar: How to Choose the Best?...
- S Style and Beauty How to Get Rid of a Bruise: Tips and Tricks...
- H Health and Medicine Is Massage Necessary? Facts and Opinions...
- L Leisure and Entertainment Should You Buy a Ceramic Knife?...
- C Computers and Internet Best Antivirus: How to Choose Protection for Your Computer?...
Answers on questions: Chemistry
- C Chemistry The formation of the iron(III) thiocyanate complex ion is an exothermic equilibrium system. For each change to the system, indicate how the concentration of each species...
- C Chemistry Steam reforming of methane ( ) produces synthesis gas, a mixture of carbon monoxide gas and hydrogen gas, which is the starting point for many important industrial chemical...
- C Chemistry The amount of a harmful chemical to which a person is exposed is the a. dose b. response c. pollutant d. toxin...
- C Chemistry What steps are performed when naming ionic compounds?...
- C Chemistry Dillon shakes a bottle of oil and vinegar salad dressing. the dressing mixes temporarily, but does not stay mixed. what property of the ingredients makes them separate?...
- C Chemistry Astudent made the following observations about astronomical bodies. observation 1: mars reverses its direction of motion in the sky. observation 2: the stars appear brighter...
- C Chemistry The kinetic energy of a moving object depends on its mass and its a. volume. b. velocity. c. distance. d. acceleration....
- C Chemistry How does the atmosphere make Earth warmer?...
- C Chemistry Fill in the blanks i will mark brainlist and 20 points i need help asap plsss...
- C Chemistry A hypothesis is to explain a set of observations. 1 one possible way 2 the correct method 3 a factual statement4 a set of data...

Ответ:
Option (d) is the correct answer.
Explanation:
The given data is as follows.
n = 0.111 mole, T = 1130 degrees Celsius = 1130 + 273 = 1403 K
P = 5.45 atm, V = ?
R = 0.0821 L atm/mol K
Using the ideal gas equation, we can calculate the volume as follows.
PV = nRT
or, V =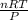
=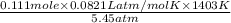
=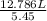
= 2.346 L
or, = 2.35 L (approx)
Thus, we can conclude that volume of the ideal gas is 2.35 liters.
Ответ:
pH = f(x) = -log₁₀x
1. Graphs
I used Excel to calculate the pH values and draw the graphs (see the Figure).
f(x) and f(x) +1 are plotted against the left-hand axis, while f(x+ 1) is plotted against the expanded right-hand axis.
The points at which pH = 0 and pH = 1 are indicated by the large red dots.
2. x = 0.5
When x = 0.5, pH ≈0.30. The point is indicated by the red diamond.
3. Transformations
(a) ƒ(x) = -log(x) + 1
This function has no y-intercept, because log(0) is undefined.
(b) ƒ(x +1) = -log(x + 1)
f(0) = -log(0 + 1) = -log(1) = 0
This function has a y-intercept at (0,0).
hope this helps please mark me brainliest!