Kingmoney959
04.01.2020 •
Chemistry
Which ion is formed when an atom of fluorine (f) gains one electron?
f–1
f–2
f+1
f+2
Solved
Show answers
More tips
- L Leisure and Entertainment Learn How to Knit with Needles: A Simple Guide for Beginners...
- C Computers and Internet Best Antivirus: How to Choose Protection for Your Computer?...
- L Legal consultation What Documents Are Required for a Russian Passport?...
- S Sport How to Choose Tennis Rackets?...
- H Health and Medicine AKDS Vaccination: Ensure Your Child s Safety...
- H Health and Medicine Naskol ko Opasen Ukus Kleshcha i Kak Ego Raspoznat...
- C Computers and Internet How to Delete Your Account on Odnoklassniki...
- H Health and Medicine What to Do When Your Jaw Locks Up?...
- G Goods and services What Are the Most Popular Services?...
Answers on questions: Chemistry
- P Physics Which of the following is correct technique for performing a push up? a. waist bent at the hips b. neck bent c. head, hips, and feet form a straight line d. none of the above...
- E English Do you think modeling is useful technique for a beginning writer...
- C Chemistry Which word or phrase below best describes the function of the urinary bladder? a. filter b. storage tank c. blender d. passageway...
- C Chemistry When a cell is in a solution where the concentration of solute is the same in the cell as in the solution, the solution is called?...
- C Chemistry Graphing Independent and Dependent Variables...

Ответ:
Ответ:
Option (a) is the correct answer.
Explanation:
When an atom or element gains an electron then the atom acquires a negative charge whereas when an atom or element loses an electron then it acquires a positive charge.
for example, atomic number of fluorine is 9 and when it gains one electron then fluorine atom acquires a negative charge. Hence, neutral atom of fluorine changes into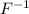 ion.
ion.
Thus, we can conclude that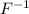 ion is formed when an atom of fluorine (F) gains one electron.
ion is formed when an atom of fluorine (F) gains one electron.
Ответ: