Which of the following is not a pure substance?
Solved
Show answers
More tips
- H Health and Medicine Simple and Effective: How to Get Rid of Cracked Heels...
- O Other How to Choose the Best Answer to Your Question on The Grand Question ?...
- L Leisure and Entertainment History of International Women s Day: When Did the Celebration of March 8th Begin?...
- S Style and Beauty Intimate Haircut: The Reasons, Popularity, and Risks...
- A Art and Culture When Will Eurovision 2011 Take Place?...
- S Style and Beauty How to Choose the Perfect Hair Straightener?...
- F Family and Home Why Having Pets at Home is Good for Your Health...
- H Health and Medicine How to perform artificial respiration?...
- H Health and Medicine 10 Tips for Avoiding Vitamin Deficiency...
- F Food and Cooking How to Properly Cook Buckwheat?...
Answers on questions: Chemistry
- C Chemistry In which cellular structure is food produced in plants...
- C Chemistry How many moles of H2O can you make from the sulfuric acid? Use the following balanced equation: H2SO4 + 2NaOH -- 2H2O + Na2SO4...
- C Chemistry el número músico representa la suma del número de protones (z) y del mimero de eletrones (n) existe en el núcleo de un automo...
- C Chemistry Plz Hurry This Is TimedThank You...
- E English Drag and drop each word into the category that describes its connotation intelligent Positive Negative confident angry brainy antique old fashioned concerned pushy...
- M Mathematics Choose the slope-intercept form of 3x + 2y = 5. 3. у==x 5 2. O O y=-x+ 5 0 5 v=-x+ O yox 5 3...
- M Mathematics What is the equation of the line that passes through (-12,6) and (-6,1)?...
- M Mathematics Select all the expressions that represent the difference of the length to width ratios for rectangle A and rectangle B, where W is the width of rectangle A....
- G Geography Which is a true statement about the diagram?...
- B Biology what is the equation for cellular respiration? and which items are the reactants and which items are the products?...

Ответ:
Ответ:
The enthalpy of the reaction is coming out to be -902 kJ.
Explanation:
Enthalpy change is defined as the difference in enthalpies of all the product and the reactants each multiplied with their respective number of moles. It is represented as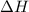
The equation used to calculate enthalpy change is of a reaction is:
For the given chemical reaction:
The equation for the enthalpy change of the above reaction is:
We are given:
Putting values in above equation, we get:
Hence, the enthalpy of the reaction is coming out to be -902 kJ.