Which substance yields hydroxide ion as the only negative ion in aqueous solution?
Solved
Show answers
More tips
- F Food and Cooking What s the Best Rice for Cooking Plov?...
- F Family and Home How to Remove Fading from Clothes: Tips and Tricks...
- F Food and Cooking How to Make Polendwitsa at Home?...
- F Family and Home Parents or Environment: Who Has the Most Influence on a Child s Upbringing?...
- P Philosophy Unbelievable stories of encounters with otherworldly forces...
- L Leisure and Entertainment How to Choose the Perfect Gift for Men on February 23rd?...
- H Health and Medicine How to Treat Whooping Cough in Children?...
- H Health and Medicine Simple Ways to Lower Cholesterol in the Blood: Tips and Tricks...
- O Other How to Choose the Best Answer to Your Question on The Grand Question ?...
- L Leisure and Entertainment History of International Women s Day: When Did the Celebration of March 8th Begin?...
Answers on questions: Chemistry
- B Business In 2010, $1.00 U.S. bought 8.24 Chinese yuan and in 2012 it bought 6.64 Chinese yuan. How many U.S. dollars could 1 Chinese yuan purchase in 2010 and 2012? a.2010: .12 U.S....
- C Chemistry What is the volume of a diamond with a mass of 0.25 g?...
- P Physics Acurrent of 3 a is running through a thin gold strip that is applied to an external magnetic field of 0.700 t. the length of the gold strip in the direction of the current is...
- B Business Q 6.3: Mia received a credit card offer in the mail. The credit card has an annual percentage rate of 26%. What is the approximate monthly interest rate that will apply to any...
- B Business Explain 5 benefits why you should keep your business small...

Ответ:
a. Mg(OH)2.
b. CH3Cl.
c. MgCl2 .
d. C2H4(OH)2.
Answer is: a. Mg(OH)₂.
An Arrhenius base is a substance that dissociates in water to form hydroxide ions (OH⁻).
In this example magnesium hydroxide is an Arrhenius base:
Mg(OH)₂(aq) → Ba²⁺(aq) + 2OH⁻(aq).
CH₃Cl and C₂H₄(OH)₂ are organic compound and will not give hydroxide ions in water, MgCl₂ is neutral salt and it will give Cl⁻ ions.
Ответ:
-
12.41 g
Explanation: -
Mass of CO₂ = 42 g
Molar mass of CO₂ = 12 x 1 + 16 x 2 = 44 g / mol
Number of moles of CO₂ =
= 0.9545 mol
The balanced chemical equation for this process is
2C₆H₆ + 15O₂ → 12CO₂ + 6H₂O
From the balanced chemical equation we see
12 mol of CO₂ is produced from 2 mol of C₆H₆
0.9545 mol of CO₂ is produced from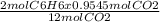
= 0.159 mol of C₆H₆
Molar mass of C₆H₆ = 12 x 6 + 1 x 6 =78 g /mol
Mass of C₆H₆ =Molar mass x Number of moles
= 78 g / mol x 0.159 mol
= 12.41 g