Why must a solution of naoh be standardized using a primary standard, such as khp?
Solved
Show answers
More tips
- F Food and Cooking How to Make Napoleon Cake: A Step-by-Step Guide...
- H Health and Medicine How to Increase Hemoglobin in the Blood...
- F Food and Cooking How to Make Chebureki: A Delicious Recipe for Russian Street Food...
- S Style and Beauty Ultimate Guide on How to Care for Suede Shoes...
- T Travel and tourism How to Use a Compass: A Beginner s Guide...
- D Dating, Love, Relationships Does a Person s Character Depend on the Color of Their Eyes?...
- F Food and Cooking Is it Really Possible to Cook Tasty Colored Cauliflower?...
- H Health and Medicine Why do our Joints Crack?...
- H Health and Medicine These Food Additives Can Do Significant Harm to Your Health...
- F Food and Cooking How to Choose the Right Olive Oil: A Comprehensive Guide...
Answers on questions: Chemistry
- C Chemistry Which layer is the thinnest? A.Mantle B.Oceanic Crust C.Continental Crust D.Outer Core...
- C Chemistry All Matter has properties. Properties are characteristics that we can use to tell them apart....
- C Chemistry What phase is calcium at 1,000K?...
- C Chemistry 2 Which property is always the same before and after a chemical reaction? A mass B volume density D color Hurry plz help me ‼️...
- C Chemistry Whats the answer to this chemisrty question...
- C Chemistry How many molecules is 3.45 grams of BrCl? ...
- C Chemistry Plants have many specialized structures that are required for sexual reproduction. Is this true in other organisms as well?” Provide an explanation with data and reasoning...
- C Chemistry Which pair of elements has the most similar chemical properties? a. P and As b. As and Se c. C and F d. Li and Be...
- C Chemistry Your lungs take in air through the mouth and nose. Explain how the lungs function in the human body. They pass oxygen from the air into the blood. They remove carbon dioxide...
- C Chemistry 69755.7 mm Hg. The average temperature is 467 °C. Assuming 1 mole of air has a volume of 22.41 Liters, what is the gas constant, R, on Venus?...

Ответ:
Answer : The molecule likely to be planar is,
Explanation :
Formula used :
where,
V = number of valence electrons present in central atom
N = number of monovalent atoms bonded to central atom
C = charge of cation
A = charge of anion
Now we have to determine the hybridization of the following molecules.
(A) The given molecule is,
The number of electron pair are 5 that means the hybridization will be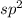 and the electronic geometry of the molecule will be trigonal planar.
and the electronic geometry of the molecule will be trigonal planar.
(B) The given molecule is,
The number of electron pair are 4 that means the hybridization will be and the electronic geometry of the molecule will be tetrahedral.
and the electronic geometry of the molecule will be tetrahedral.
But as there are 3 atoms around the central oxygen atom, the fourth position will be occupied by lone pair of electrons. The repulsion between lone and bond pair of electrons is more and hence the molecular geometry will be pyramidal.
(C) The given molecule is,
The number of electron pair are 4 that means the hybridization will be and the electronic geometry of the molecule will be tetrahedral.
and the electronic geometry of the molecule will be tetrahedral.
But as there are 2 atoms around the central oxygen atom, the fourth position will be occupied by lone pair of electrons. The repulsion between lone and bond pair of electrons is more and hence the molecular geometry will be pyramidal.
(d) The given molecule is,
The number of electron pair are 4 that means the hybridization will be and the electronic geometry of the molecule will be tetrahedral.
and the electronic geometry of the molecule will be tetrahedral.
(E) The given molecule is,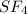
The number of electron pair are 5 that means the hybridization will be and the electronic geometry of the molecule will be tetrahedral.
and the electronic geometry of the molecule will be tetrahedral.
But as there are 4 atoms around the central oxygen atom, the fifth position will be occupied by lone pair of electrons. The repulsion between lone and bond pair of electrons is more and hence the molecular geometry will be sea-saw.
Hence, the molecule likely to be planar is,