Write the net ionic equation for the reaction that occurs when equal volumes of 0.062 M aqueous formic acid and sodium benzoate are mixed. It is not necessary to include states such as (aq) or (s). Use HCOO- as the formula for the formate ion.
Solved
Show answers
More tips
- F Family and Home How to Properly Use a Water Level?...
- D Dating, Love, Relationships 10 Useful Tips on How to Survive a Breakup?...
- F Food and Cooking Apple Cider Vinegar: The Ultimate Health and Beauty Solution...
- C Computers and Internet Е-head: How it Simplifies Life for Users?...
- F Family and Home How to Choose the Best Diapers for Your Baby?...
- F Family and Home Parquet or laminate, which is better?...
- L Leisure and Entertainment How to Properly Wind Fishing Line onto a Reel?...
- L Leisure and Entertainment How to Make a Paper Boat in Simple Steps...
- T Travel and tourism Maldives Adventures: What is the Best Season to Visit the Luxurious Beaches?...
- H Health and Medicine Kinesiology: What is it and How Does it Work?...
Answers on questions: Chemistry
- M Mathematics PLZ HELP, I WILL MARK U BRAINLLEST write three equivalent decimal forms for the number 1.1 repeating...
- M Mathematics Which quadratic function is shown in the graph a) f(x)= x^2-2x+3 b) f(x)= x^2-3x+2 c) f(x)= -x^2-2x+3 d) f(x)= -x^2-3x+2...
- H Health True/False: 11. Touching a ground ball with the hand or arms is a violation?...

Ответ:
The net ionic equation when formic acid and sodium benzoate are mixed is as follows.
Explanation:
A chemical equation in which both reactants and products are present in the form of ions is called an ionic equation.
The chemical formula of formic acid is HCOOH and chemical formula of sodium benzoate is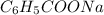 .
.
Therefore, net ionic equation when formic acid and sodium benzoate are mixed is as follows.
Ответ:
volume HCl = 3.4 g hcl ( 1 mol hcl / 36.46 g hcl) (1 L / 0.5 mol hcl )
volume HCl = 0.1865 L