novarosell
16.03.2020 •
Chemistry
Write the precipitation reaction for calcium chloride in aqueous solution: Use the pull-down menus to specify the state of each reactant and product. CaCl2 + Is calcium chloride considered soluble or not soluble ? ... ... A. Soluble ... B. Not soluble
Solved
Show answers
More tips
- F Food and Cooking The Health Benefits of Flaxseed oil...
- S Style and Beauty Why is Sugaring Better than Waxing for Hair Removal?...
- W Work and Career Where can you learn to be a flight attendant?...
- G Goods and services How to Properly Calculate the Power of Your Air Conditioner?...
- F Food and Cooking Effective Methods to Organize Videos in your iPad According to Content...
- F Family and Home Parquet or laminate, which is better?...
- L Leisure and Entertainment How to Properly Wind Fishing Line onto a Reel?...
- L Leisure and Entertainment How to Make a Paper Boat in Simple Steps...
- T Travel and tourism Maldives Adventures: What is the Best Season to Visit the Luxurious Beaches?...
- H Health and Medicine Kinesiology: What is it and How Does it Work?...
Answers on questions: Chemistry
- M Mathematics Gebhardt Electronics produces a wide variety of transformers that it sells directly to manufacturers of electronics equipment. For one component used in several models of its...
- S Social Studies How long would it take you to save up to $1000 if you put back $20 a week?...
- B Business All of the five elements of manufacturing contribute to the cost of production. which one of the following is not one of the 5ms? a. methods b. mandates c. measurement d. machinery...
- M Mathematics A15 foot rope stretches diagonally from the ground to the top of a tent 9 feet off the ground. what angle does the rope form with the ground?...

Ответ:
Option (A) is the correct answer.
Explanation:
It is known that like dissolves like. So, water being a polar solvent is able to dissolve ionic compounds in it.
For example,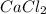 is an ionic compound as it's atoms contain a metal and a non-metal.
is an ionic compound as it's atoms contain a metal and a non-metal.
Therefore, the bond present present in it is an ionic bond. Hence, when it is dissolved in water then its reaction equation is as follows.
Thus, we can conclude that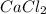 is soluble.
is soluble.
Ответ:
Density = 1.504 g/mL⁻¹
Molar mass HNO₃ =63.01 g/mol
M = 1000 * T* D / mm
M = 1000 * 0.68 * 1.504 / 63.01
M = 1022.72 / 63.01
M = 16.23 mol/L
hope this helps!