hellothere2458
26.10.2020 •
Geography
How far is Uranus from the sun in km?
Solved
Show answers
More tips
- C Computers and Internet Log-in: what is it and why do you need it?...
- S Style and Beauty Secrets of Tying a Pareo: 5 Ways...
- F Food and Cooking 10 Reasons Why You Should Avoid Giving Re-Gifts: An Informative Guide...
- S Sport How to wrap boxing hand wraps? Everything you need to know!...
- A Animals and plants 5 Tips for Taking Care of Yews to Keep Them Green and Beautiful...
- H Health and Medicine How to Calm Your Nerves? Expert Tips That Actually Work...
- O Other What is a Disk Emulsifier and How Does it Work?...
- S Sport How to Pump Your Chest Muscle? Secrets of Training...
- C Computers and Internet How to Get Rid of 3pic Infector: Everything You Need to Know...
- S Style and Beauty How to Grow Hair Faster: Real Methods and Advice...
Answers on questions: Geography
- L Law How did the Supreme Court s McCulloch v. Maryland decision support the Constitution s Necessary and Proper Clause as well as the Supremacy Clause?...
- M Mathematics Please help quickly?????...
- P Physics At a certain time a particle had a speed of 48 m/s in the positive x direction, and 4.5 s later its speed was 92 m/s in the opposite direction. What was the average acceleration...

Ответ:
2.959 billion km away
Explanation:
I don't really know the explanation besides the distance is large because it is at a very high orbit but other than that I looked it up. (Just multiply 2.959 by 1 billion to get the full number)
Ответ:
Explanation: Uranus is about 19.8 astronomical units away from the sun.
Ответ:
Explanation:
Given parameters:
Mass of the sample = 0.622g
Volume of the gas = 2.4L
Temperature = 287K
Pressure of the gas = 0.85atm
Unknown
Molar mass of the gas = ?
Solution
To find the molar mass of the gas, we first assume that the gas in question is an ideal gas i.e. it obeys Boyle's and Charles's law.
On this premise, we can find the unknown which is the molar mass of the gas.
We first use the ideal gas equation to find the number of moles of the gas since all the parameters apart from the number of moles are given:Solving for number of moles:
The ideal gas equation is given as PV = nRT
We make the unknown "n" the subject of the equation
n =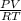
R is the gas constant whose value is 0.082atmdm³mol⁻¹K⁻¹
n =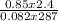
n =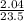 = 0.087mole
= 0.087mole
Now, we know the number of moles of the gas. We can proceed to find the molar mass from the given mass and the number of moles calculated using the equation below:molar mass =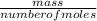
molar mass =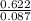 =7.15gmol⁻¹
=7.15gmol⁻¹
Part 2:
If the sample was placed under a low temperature, we want to consider what would happen to the volume:
Since we assume that the gas is an ideal gas, we can explain what will happen to the gas using Charles's law. The law states that "The volume of a fixed mass of a gas varies directly as its absolute temperature if the pressure is constant".
If we assume the pressure is constant for this gas, the volume of the gas would decrease as the temperature becomes lower. We would get lower values for both actual volume and predicted volume.