lovelife132015
12.02.2021 •
Physics
A bottle of water with mass 0.9 kg is left out in the sun, the radiation from the sun warms up the water bottle. If the water bottle was initially 10°C and the sun provided 120,000 J of thermal energy, calculate the final temperature of the water. Use C = 4182 j/kgK for the specific heat of water.
Solved
Show answers
More tips
- H Health and Medicine When can it be said that a person has a normal pulse?...
- A Art and Culture When Will Eurovision 2011 Take Place?...
- S Style and Beauty How to Choose the Perfect Hair Straightener?...
- F Family and Home Why Having Pets at Home is Good for Your Health...
- H Health and Medicine How to perform artificial respiration?...
- H Health and Medicine 10 Tips for Avoiding Vitamin Deficiency...
- F Food and Cooking How to Properly Cook Buckwheat?...
- F Food and Cooking How Many Grams Are In a Tablespoon?...
- L Leisure and Entertainment Carving: History and Techniques for Creating Vegetable and Fruit Decorations...
- P Photography and Videography How to Choose the Perfect Photo Paper for Your Images?...
Answers on questions: Physics
- P Physics Two long, parallel wires are separated by a distance of 2.00 cm . The force per unit length that each wire exerts on the other is 3.60×10^−5 N/m , and the wires repel each other....
- P Physics During the pitching motion, a baseball pitcher exerted an average horizontal force of 60 N against the 0.15 kg baseball while moving it through a horizontal displacement of...
- P Physics A point charge of 6.0 nC is placed at the center of a hollow spherical conductor (inner radius = 1.0 cm, outer radius = 2.0 cm) which has a net charge of –4.0 nC. Determine...
- P Physics This baeeeeeeeeeeeeeeeeee...
- P Physics Need help finding the average speed....
- P Physics While researching life during colonial times, carson learns that people would extract potassium hydroxide from wood ashes and mix it with animal fat. what were the colonists...
- M Mathematics What is the factored form of the expression? w^2+16w+64 a(w+8)(w-8) b(w+64)(w-1) c(w+8)^2 d(w-8)^2...
- H Health Which statements about the warming effects of sunlight are true? on a sunny day, a black leather car seat would be hotter than a tan cloth seat. the sun s rays interact the...
- G Geography Which angle is a vertical angle with angleefd? a. angledfb b. angleced c. anglebfc d. angleafb...
- M Mathematics Solve for x.. a) x= 8 b) x= 22.5 c) x= 32.4 d) x= 40.5...

Ответ:
Final temperature, T2 = 314.9 Kelvin
Explanation:
Given the following data:
Mass = 0.9kg
Initial temperature, T1 = 10°C to Kelvin = 10 + 273 = 283K
Quantity of heat = 120,000 J
Specific heat capacity = 4182 j/kgK
To find the final temperature;
Heat capacity is given by the formula;
Where;
Q represents the heat capacity or quantity of heat.
m represents the mass of an object.
c represents the specific heat capacity of water.
dt represents the change in temperature.
Making dt the subject of formula, we have;
Substituting into the equation, we have;
dt = 31.9K
Now, the final temperature T2 is;
But, dt = T2 - T1
T2 = dt + T1
T2 = 31.9 + 283
T2 = 314.9 Kelvin
Ответ:
Explanation:
It is given that,
Mass of the parachutist, m = 100 kg
It falls from a height of 500 meters.
When the parachutist reaches the ground, the potential energy gets converted to its kinetic energy. The potential energy of the parachutist is given by the following formula as :
g is the acceleration due to gravity
Since, 1 kJ = 1000 J
So,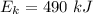
So, as the parachutist reaches ground its kinetic energy is equal to 490 kJ.