A gas is compressed at constant temperature from a volume of 5.68 L to a volume of 2.35 L by an external pressure of 732 torr. Calculate the work done in J
Solved
Show answers
More tips
- O Other How to Choose the Best Answer to Your Question on The Grand Question ?...
- L Leisure and Entertainment History of International Women s Day: When Did the Celebration of March 8th Begin?...
- S Style and Beauty Intimate Haircut: The Reasons, Popularity, and Risks...
- A Art and Culture When Will Eurovision 2011 Take Place?...
- S Style and Beauty How to Choose the Perfect Hair Straightener?...
- F Family and Home Why Having Pets at Home is Good for Your Health...
- H Health and Medicine How to perform artificial respiration?...
- H Health and Medicine 10 Tips for Avoiding Vitamin Deficiency...
- F Food and Cooking How to Properly Cook Buckwheat?...
- F Food and Cooking How Many Grams Are In a Tablespoon?...
Answers on questions: Physics
- P Physics What type of forces are needed in a system for the object to move?...
- P Physics Please help me understand this question and the process of how to do it!!! (I need step by step also)...
- P Physics a stone is released from rest at a height of 3m above the ground. it accelerates uniformly under the earth’s gravitational pull at 10m/s^2. at what speed does the...
- P Physics Please help it’s due tomorrow...
- P Physics . State any three applications of conduction...
- P Physics Explain the flow of energy needed to listen to music through headphones....
- P Physics 50cm3 of 1.5M ammonia solution were shaken with 50cm3 of Chloroform in separating funnel. After allowing the layer to settle 20cm3 of the chloroformlayer were with...
- P Physics Please helpp!!! What is true about a projectile motion? a) A projectile moves through air or space b) A projectile is subject to the force of gravity c) A projectile...
- M Mathematics Find 5 fractions that equal to 1/5...
- M Mathematics 4.8 as an improper fraction...

Ответ:
The work done in J is 324
Explanation:
To calculate the amount of work done for an isothermal process is given by the equation:
W = amount of work done = ?
P = pressure = 732 torr = 0.96 atm (760torr =1atm)
Putting values in above equation, we get:
To convert this into joules, we use the conversion factor:
So,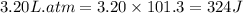
The positive sign indicates the work is done on the system
Hence, the work done for the given process is 324 J
Ответ:
Hope that helped:)