narwhalebearp5871i
13.04.2021 •
Physics
The temperature of a 773-g block of ice increases from -15.62°C to -13.71°C. How much heat, in joules, was absorbed by the block of ice? (The specific heat of ice is 2.1 J/g • °C.)
Solved
Show answers
More tips
- W Work and Career How to Start Your Own Business: Tips and Recommendations...
- S Society and Politics 10 Tips for Boosting Your Self-Esteem...
- C Computers and Internet How to Create a Folder on Your iPhone?...
- G Goods and services How to sew a ribbon: Tips for beginners...
- F Food and Cooking How to Make Mayonnaise at Home? Secrets of Homemade Mayonnaise...
- C Computers and Internet Which Phone is Best for Internet Surfing?...
- F Food and Cooking Everything You Need to Know About Pasta...
- C Computers and Internet How to Choose a Monitor?...
- H Horoscopes, Magic, Divination Where Did Tarot Cards Come From?...

Ответ:
Compound
Explanation:
Element is a pure substance which is composed of atoms of similar elements.It can not be decomposed into simpler constituents using chemical reactions.Example: Copper
Compound is a pure substance which is made from atoms of different elements combined together in a fixed ratio by mass.It can be decomposed into simpler constituents using chemical reactions. Example: water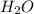
Thus as it give that the red sample on being heated, a gas is released and a blue powder is left after the heating. Thus the given sample is a compound.