edeliz3886
28.03.2020 •
Chemistry
A 63.5 g sample of an unidentified metal absorbs 355 ) of heat when its temperature changes
by 4.56 °C. Calculate the specific heat capacity of the metal.
Solved
Show answers
More tips
- F Food and Cooking Discover Delicious Recipes You Can Make with Ground Meat...
- C Computers and Internet Google Search Tips and Tricks: Everything You Need to Know...
- S Science and Technology Why is there no gravity on other planets?...
- L Leisure and Entertainment How to Properly Wind Fishing Line onto a Reel?...
- L Leisure and Entertainment How to Make a Paper Boat in Simple Steps...
- T Travel and tourism Maldives Adventures: What is the Best Season to Visit the Luxurious Beaches?...
- H Health and Medicine Kinesiology: What is it and How Does it Work?...
- O Other How to Choose the Best Answer to Your Question on The Grand Question ?...
- L Leisure and Entertainment History of International Women s Day: When Did the Celebration of March 8th Begin?...
- S Style and Beauty Intimate Haircut: The Reasons, Popularity, and Risks...
Answers on questions: Chemistry
- C Chemistry Approximately what percentage of the world s rock record is attributed to turbidity currents?...
- C Chemistry Wou would silver react with dilute sulfuric acid why or why not...
- C Chemistry Would magnesium react with dilute sulfuric acid why or why not...
- C Chemistry Which is a biological effect of low exposure to radiation? a. dna damage b. cell, tissue, and organ death c. death of the organism d. acute radiation syndrome...
- C Chemistry When examining features on the ocean floor, like a volcano, how would it appear?...
- C Chemistry Select all that apply.the thick sedimentary rocks which are located off the continental margins may contain commercial quantities of: diamondsgasgoldpetroleum...
- C Chemistry List the symblos for the nine matalloids on the periodic table?...
- C Chemistry If you make a mistake and deliver less than 50.00 mL of HCl to the Erlenmeyer flask, what effect will this have on the calculation for the acid-neutralizing ability of the antacid?...
- C Chemistry 20 mL of an approximately 10% aqueous solution of ethylamine, CH3CH2NH2, is titrated with 0.3000 M aqueous HCl. Which indicator would be most suitable for this titration? The pKa of...
- C Chemistry 1.0 g of a protein is dissolved in 100 ml of distilled water. The resulting solution has an osmotic pressure of 0.001 atm at room temperature. What is the molecular weight of the protein?...

Ответ:
0.208 is the specific heat capacity of the metal.
Explanation:
Given:
mass (m) = 63.5 grams 0R 0.0635 kg
Heat absorbed (q) = 355 Joules
Δ T (change in temperature) = 4.56 degrees or 273.15+4.56 = 268.59 K
cp (specific heat capacity) = ?
the formula used for heat absorbed and to calculate specific heat capacity of a substance will be calculated by using the equation:
q = mc Δ T
c =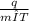
c =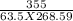
= 0.208 J/gm K
specific heat capacity of 0.208 J/gm K
The specific heat capacity is defined as the heat required to raise the temperature of a substance which is 1 gram. The temperature is in Kelvin and energy required is in joules.
Ответ: