michaelbromley9759
18.03.2020 •
Chemistry
A calorimeter contains 32.0 mLmL of water at 15.0 ∘C∘C . When 1.20 gg of XX (a substance with a molar mass of 54.0 g/molg/mol ) is added, it dissolves via the reaction X(s)+H2O(l)→X(aq)X(s)+H2O(l)→X(aq) and the temperature of the solution increases to 29.0 ∘C∘C . Calculate the enthalpy change, ΔHΔHDelta H, for this reaction per mole of XX. Assume that the specific heat of the resulting solution is equal to that of water [4.18 J/(g⋅∘C)J/(g⋅∘C)], that density of water is 1.00 g/mLg/mL, and that no heat is lost to the calorimeter itself, nor to the surroundings.
Solved
Show answers
More tips
- F Family and Home Parquet or laminate, which is better?...
- L Leisure and Entertainment How to Properly Wind Fishing Line onto a Reel?...
- L Leisure and Entertainment How to Make a Paper Boat in Simple Steps...
- T Travel and tourism Maldives Adventures: What is the Best Season to Visit the Luxurious Beaches?...
- H Health and Medicine Kinesiology: What is it and How Does it Work?...
- O Other How to Choose the Best Answer to Your Question on The Grand Question ?...
- L Leisure and Entertainment History of International Women s Day: When Did the Celebration of March 8th Begin?...
- S Style and Beauty Intimate Haircut: The Reasons, Popularity, and Risks...
- A Art and Culture When Will Eurovision 2011 Take Place?...
- S Style and Beauty How to Choose the Perfect Hair Straightener?...
Answers on questions: Chemistry
- G Geography The Manhattan project produced the first...
- M Mathematics What is the least common multiple of 2x^2 and 4x+12...
- H History Việt nam đã có chủ nghĩa xã hội chưa...
- B Biology chordates possess several distinguishing characteristics that separate them from other animal groups. which of these features is not found in all chordates? a. postanal tailb. pharyngeal...

Ответ:
The enthalpy change ΔH for this reaction per mole of X is -181.10 mol/kJ.
Explanation:
Volume of water in calorimeter = 32.0 mL
Mass of water in calorimeter = M
Density of the water = d = 1.00 g/mL
Mass of substance = 1.20 g
Mass of solution ,m= 1.20 g+ 32.0 g = 33.20 g
Specific heat of solution = c = 4.18 J/g°C
Change in temperature of the calorimeter = ΔT = 29.0°C
Heat absorbed by the solution = Q
Moles of substance X added to water ,n=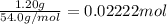
Heat released when 1.20 grams of X was dissloved = Q'= -Q = -4,024.504 J
Q' = -4,024.504 J = -4.024504 kJ ≈ 4.0245 kJ
1 J = 0.001 kJ
Enthalpy change for this reaction per mole of X:
Ответ:
Yes 91 lol but 9+1 is 10.