wildfire771003
11.08.2021 •
Chemistry
A) Define typical polyfunctional acid ?
b) Show the equations of dissociation mechanism of phosphoric acid as an example.
c) Write the equation for calculating the [H3O*].
Solved
Show answers
More tips
- C Computers and Internet Why is Yandex s robot Bless.yandex.ru Important?...
- F Food and Cooking Fresh and Easy Lemonade in 10 Minutes...
- S Sport How to wrap boxing hand wraps? Everything you need to know!...
- F Family and Home How to Sew Curtain Tapes: Best Tips from Professionals...
- A Animals and plants How to Grow Lime from a Seed: Simple Tips and Interesting Facts...
- C Computers and Internet How to Create a Folder on Your iPhone?...
- G Goods and services How to sew a ribbon: Tips for beginners...
- F Food and Cooking How to Make Mayonnaise at Home? Secrets of Homemade Mayonnaise...
- C Computers and Internet Which Phone is Best for Internet Surfing?...
- F Food and Cooking Everything You Need to Know About Pasta...
Answers on questions: Chemistry
- C Chemistry Tell me the answer to the following question and if you can explain it, if you will i ll vote your answer the brainliest and you ll receive more points. you! which of...
- C Chemistry What are the disadvantages of aluminium fir building cars...
- C Chemistry What is the molarity of a 10 l solution containing 5.0 moles of solute?...
- C Chemistry 1. How many GRAMS of copper(II) fluoride are present in 1.63 moles of this compound? grams. 2. How many MOLES of copper(II) fluoride are present in 2.38 grams of this...
- C Chemistry 1. Which of the following shows the process of freezing? A.candle wax cooling on the table B.pouring cola over ice C. heating a cup of water D. particles are speeding-up...
- C Chemistry I need a hand with this one: Having this equation: Na2SO4 +BaCl2 = BaSO4+NaCl If 10.09 grams of Na2SO4 were added to a 50mL barium solution and reacted completely, what...
- C Chemistry a lizard drinks water that contains H-2. The H-2 decays and approximately 6.25% remain after 30 seconds. What is the 1/2 life for H-2?...
- C Chemistry What is the reaction of guanine and adenine...
- C Chemistry If the percent transmittance is is 64.9 in 0.2M standard CuSO4, what is the absorption?...
- C Chemistry Aluminum + lead(ii) aluminum nitrate +lead balanced equation...

Ответ:
a) A polyfunctional acid is an acid that has more than one functional group.
b) The equations of dissociation of phosphoric acid are:
H₃PO₄ + H₂O ⇄ H₂PO₄⁻ + H₃O⁺ H₂PO₄⁻ + H₂O ⇄ HPO₄²⁻ + H₃O⁺ HPO₄²⁻ + H₂O ⇄ PO₄³⁻ + H₃O⁺c) The equation for calculating the concentration of H₃O⁺ is![[H_{3}O^{+}] = (\frac{K_{1}K_{2}K_{3}[H_{3}PO_{4}]}{[PO_{4}^{-3}]})^{1/3}](/tpl/images/1407/4133/1f59f.png)
a) A polyfunctional acid can be defined as an acid that has more than one functional group. Phosphoric acid (H₃PO₄) is an example of polyfunctional acid since it is composed of three hydroxyl groups joined to a phosphorus atom, which is also joined to an oxygen atom by a double bound. In that structure, the three hydrogen atoms of the hydroxyl groups give the acidic behavior to this compound.
b) Phosphoric acid has three equations of dissociation:
H₃PO₄ + H₂O ⇄ H₂PO₄⁻ + H₃O⁺ (1)H₂PO₄⁻ + H₂O ⇄ HPO₄²⁻ + H₃O⁺ (2)HPO₄²⁻ + H₂O ⇄ PO₄³⁻ + H₃O⁺ (3)The dissociation constants for the three above equations are:
c) We can calculate the concentration of H₃O⁺ for each equilibrium with the equations (4), (5), and (6).
The general reaction of dissociation of phosphoric acid is given by the sum of equations (1), (2), and (3):
H₃PO₄ + 3H₂O ⇄ PO₄³⁻ + 3H₃O⁺ (7)
The concentration of H₃O⁺ for the total dissociation reaction (eq 7) can be found as follows:
Where:
Hence, by knowing the dissociation constants K₁, K₂ and K₃, and the concentrations of PO₄³⁻ and H₃PO₄, the [H₃O⁺] is:
You can find more about acid dissociation constant in the link:
I hope it helps you!
Ответ:
7.98 grams of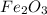 are produced if 10.7 grams of
are produced if 10.7 grams of 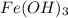 are reacted.
are reacted.
Explanation:
To calculate the number of moles, we use the equation:
Putting values in equation 1, we get:
The chemical equation for the reaction is
By Stoichiometry of the reaction:
2 moles of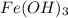 produce = 1 mole of
produce = 1 mole of 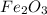
So, 0.100 moles of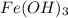 produce=
produce= 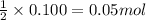 of
of 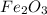
Mass of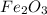 =
=
Hence 7.98 grams of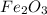 are produced if 10.7 grams of
are produced if 10.7 grams of 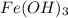 are reacted.
are reacted.