dontcareanyonemo
02.03.2020 •
Chemistry
A stock phenolphthalein solution was prepared by dissolving 0.050 g in a total volume of 100.0 ml. You place one drop (0.050 ml) of this stock solution into a flask and dilute to 100.0 ml. The measured absorbance of this diluted sample is 0.18. What is the value of molar absorptivity, ε?
Solved
Show answers
More tips
- T Travel and tourism Maldives Adventures: What is the Best Season to Visit the Luxurious Beaches?...
- H Health and Medicine Kinesiology: What is it and How Does it Work?...
- O Other How to Choose the Best Answer to Your Question on The Grand Question ?...
- L Leisure and Entertainment History of International Women s Day: When Did the Celebration of March 8th Begin?...
- S Style and Beauty Intimate Haircut: The Reasons, Popularity, and Risks...
- A Art and Culture When Will Eurovision 2011 Take Place?...
- S Style and Beauty How to Choose the Perfect Hair Straightener?...
- F Family and Home Why Having Pets at Home is Good for Your Health...
- H Health and Medicine How to perform artificial respiration?...
- H Health and Medicine 10 Tips for Avoiding Vitamin Deficiency...
Answers on questions: Chemistry
- C Chemistry Apackage of aluminum foil is 63.2 yd long, 11 in. wide, and 0.00035 in. thick. if aluminum has a density of 2.70 g/cm³, what is the mass, in grams, of the foil?...
- C Chemistry What is the minimum number of moles of sodium borohydride required to fully reduce 0.55 grams of benzophenone? enter only the number with two significant figures....
- C Chemistry 102.1 g of aluminum nitrite and 174.3 g of ammonium chloride react to form aluminum chloride, nitrogen, and water. how many grams of the excess reagent remains after the reaction?...
- C Chemistry Tropospheric ozone is rapidly consumed in many reactions, including: so3(g) + no(g) no2(g) + o2(g) use the following data to calculate the instantaneous rate of the preceding reaction...
- C Chemistry What volume of propane (c3h8) is required to produce 165 liters of water according to the following reaction? (all gases are at the same temperature and pressure.) propane (c3h8)...
- C Chemistry Winston predicts that certain people will be vaporized and that certain people will never be vaporized. who? why?...
- C Chemistry Asample has a mass of 7.412 grams and a volume of 0.217 liters. what is the density of this sample?...
- C Chemistry In compounds, elements are combined in fixed proportions. What does this mean? Use an example to illustrate your point...
- C Chemistry calculate the mass of Ag3AsO4 that would be produced if enough AgNO3 solution is added to precipitate all of the AsO43- in the 100.0 mL of the 0.1 M Na3AsO4 solution. Record this...
- C Chemistry Identify the type of solid from the description. Select from the following choices: metallic solid, network solid, molecular solid, ionic solid, or amorphous solid...

Ответ:
the value of molar absorptivity is 229000
Explanation:
given data
phenol phthalein solution = 0.050 g
total volume = 100.0 ml
dilute = 100.0 ml
diluted sample = 0.18
solution
we get here concentration that is express as
concentration = ( mass of solute × 1000 ) ÷ ( molar mass of solute × volume of solution ) .............1
put here value
concentration =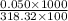
concentration = 0.00157 M
and here dillution equation is express as
c1 × v1 = c2 × v2 .................2
here c1 and c2 is initial and final concentration
and v1 and v2 is initial and final volume
put here value
0.001571 × 0.050 = c2 × 100
c2 = 7.855 ×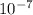 M
M
and
now we get molar by absorbance equation that is
A = E × C × l ................3
here A is absorbance and E is molar and c is absorptivity and l is path length
put here value
0.18 = E × 7.855 ×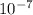 × 1
× 1
E = 229000
Ответ:
nope nope even tho idk what that mean
Explanation: