averagegirl27
13.04.2020 •
Chemistry
A student uses 100 mL of acetic acid. after the experiment the amount of acid is 1200mL with Mallory 1.5 M is formed. What is the molarity of the solution did the student used
Solved
Show answers
More tips
- H Horoscopes, Magic, Divination Where Did Tarot Cards Come From?...
- S Style and Beauty How to Make Your Lips Fuller? Ideas and Tips for Beautiful Lips...
- S Style and Beauty How are artificial nails removed?...
- F Family and Home How to Sew Curtain Tapes: Best Tips from Professionals...
- H Horoscopes, Magic, Divination How to Cast a Love Spell on a Guy? Guide for Guys...
- F Family and Home How to Properly Use a Water Level?...
- L Legal consultation What Documents Are Required for a Russian Passport?...
- H Health and Medicine How to Treat Styes: Causes, Symptoms, and Home Remedies...
- F Family and Home Protect Your Home or Apartment from Pesky Ants...
- T Travel and tourism Lost in the Catacombs: What to Do?...

Ответ:
18M
Explanation:
Given parameters:
Initial volume of acetic acid = 100mL
Final volume of solution = 1200mL = 1.2L
Final molarity = 1.5M
Unknown:
Initial molarity = ?
Solution:
We might desired a particular concentration of a compound from the stock. This way, we dilute using the right amount of solvent. One critical thing to note is that the number of moles in both solution remains the same.
To solve this problem, let us find the number of moles in the final solution.
Number of moles in final solution = molarity x volume
= 1.5 x 1.2
= 1.8mol
Now original molarity;
Since Molarity =
Molarity =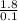 = 18M
= 18M
Ответ:
Nursing and Health also because they identify research questions and collect data and report there findings .
Physics because they find everything out and help us get to we’re we are .