aliami0306oyaj0n
16.01.2020 •
Chemistry
Achemist prepares a solution of copper(ii) fluoride (cuf2) by measuring out 0.032 µmol of copper(ii) fluoride into a 500 ml volumetric flask and filling the flask to the mark with water.calculate the concentration in mol/l of the chemist's copper(ii) fluoride solution.
Solved
Show answers
More tips
- A Auto and Moto How many blood alcohol level units are allowed in Russian traffic laws?...
- G Goods and services Kogda zhdatt Iphone 5? The Latest News and Rumors...
- F Family and Home Parquet or laminate, which is better?...
- L Leisure and Entertainment How to Properly Wind Fishing Line onto a Reel?...
- L Leisure and Entertainment How to Make a Paper Boat in Simple Steps...
- T Travel and tourism Maldives Adventures: What is the Best Season to Visit the Luxurious Beaches?...
- H Health and Medicine Kinesiology: What is it and How Does it Work?...
- O Other How to Choose the Best Answer to Your Question on The Grand Question ?...
- L Leisure and Entertainment History of International Women s Day: When Did the Celebration of March 8th Begin?...
- S Style and Beauty Intimate Haircut: The Reasons, Popularity, and Risks...
Answers on questions: Chemistry
- B Business A goal programming problem had two goals (with no priorities assigned). Goal number 1 was to achieve a profit of $2,400 and goal number 2 was to have no idle time for workers...
- H History (i can’t figure out the answer to this question and the assignment is due tomorrow)why did the founders of america think that it was necessary to limit he power of the government...

Ответ:
Answer : The concentration of copper(II) fluoride in the solution is,
Explanation : Given,
Moles of copper(II) fluoride = 0.032 μ
mol =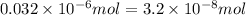
conversion used :
Volume of solution = 500 mL
Molarity : It is defined as the number of moles of solute present in one liter of volume of solution.
Formula used :
Now put all the given values in this formula, we get:
Therefore, the concentration of copper(II) fluoride in the solution is,
Ответ:
tell him how u feel about him and try to understand how he feels
Explanation: