Apolonium atom has 84 protons, 124 neutrons, and 84 electrons. what will it change to during alpha decay? a. a lead atom with 82 protons, 122 neutrons, and 84 electrons b. a bismuth atom with 83 protons, 123 neutrons, and 84 electrons c. a polonium atom with 84 protons, 124 neutrons, and 85 electrons d. an astatine atom with 85 protons, 123 neutrons, and 84 electrons
Solved
Show answers
More tips
- C Computers and Internet Protect Your Computer: How to Set a Password on Your Device...
- W Work and Career How to behave at an interview? Tips from an HR specialist...
- F Family and Home How to Keep Your Home Warm: Tips and Tricks...
- D Dating, Love, Relationships Does a Person s Character Depend on the Color of Their Eyes?...
- O Other Childhood Fears: What Many of Us Experienced...
- H Health and Medicine Simple and Effective: How to Get Rid of Cracked Heels...
- O Other How to Choose the Best Answer to Your Question on The Grand Question ?...
- L Leisure and Entertainment History of International Women s Day: When Did the Celebration of March 8th Begin?...
- S Style and Beauty Intimate Haircut: The Reasons, Popularity, and Risks...
- A Art and Culture When Will Eurovision 2011 Take Place?...
Answers on questions: Chemistry
- C Chemistry Petroleum can be separated by distillation because the hydrocarbons in petroleum are a) elements with identical boiling points b) elements with different boiling points...
- C Chemistry Which of the two aqueous solutions will be a better conductor of electricity in the following examples? explain why in each case. a)1.0m nacl vs. 0.010 m nacl b)0.10...
- C Chemistry Calculate e°cell for a silver-aluminum cell in which the cell reaction is al(s) + 3ag+(aq) → al3+(aq) + 3ag(s) –2.46 v 0.86 v –0.86 v 2.46 v...
- C Chemistry Calculate the molar solubility of baso4 in water, ksp=1.0*10^-10...
- C Chemistry Each time you increase the temperature, the solution becomes saturated supersaturated...
- C Chemistry Based on nuclear stability, what is the symbol for the most likely product nuclide when iodine-123 undergoes decay?...
- C Chemistry Calculate whether a precipitate will form (and why) if 0.35 l of 0.0044 m ca(no3)2 and 0.17 l of 0.00039 m naoh are mixed at 25◦c. ksp for ca(oh)2 is equal to 5.5 ×...
- C Chemistry Based on nuclear stability, what is the most likely product nuclide when cesium-137 undergoes decay? symbol for nuclide...
- C Chemistry Calculate the mass of sodium acetate trihydrate required to make 100. ml of a 0.100 m solution. as part of your preparation for performing this experiment, record your...
- C Chemistry Calculate the ph of sweater which has a hydrogen ion concentration of 1 times 10 to the power of 8...

Ответ:
Option (A) is the correct answer.
Explanation:
An alpha particle is a helium atom that is represented as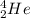 .
.
Therefore, when an alpha decay occurs if polonium atom has 84 protons, 124 mass, and 84 electrons. Then the reaction will be as follows.
An element with atomic number 82 is lead.
Thus, we can conclude that when a polonium atom has 84 protons, 124 mass, and 84 electrons then it will change to a lead atom with 82 protons, 122 mass, and 82 electrons during alpha decay.
Ответ:
i agree , a thermometer