At a certain temperature, 0.740 mol of so3 is placed in a 4.00 l container. so_{3}(g)\rightleftharpoons 2so_{2}(g)+o_{2}(g) at equilibruim, 0.190 mol of o2 is present. calculate kc.
Solved
Show answers
More tips
- T Travel and tourism Maldives Adventures: What is the Best Season to Visit the Luxurious Beaches?...
- H Health and Medicine Kinesiology: What is it and How Does it Work?...
- O Other How to Choose the Best Answer to Your Question on The Grand Question ?...
- L Leisure and Entertainment History of International Women s Day: When Did the Celebration of March 8th Begin?...
- S Style and Beauty Intimate Haircut: The Reasons, Popularity, and Risks...
- A Art and Culture When Will Eurovision 2011 Take Place?...
- S Style and Beauty How to Choose the Perfect Hair Straightener?...
- F Family and Home Why Having Pets at Home is Good for Your Health...
- H Health and Medicine How to perform artificial respiration?...
- H Health and Medicine 10 Tips for Avoiding Vitamin Deficiency...
Answers on questions: Chemistry
- C Chemistry We determined the bottom piece of tape was: a. negative, because it ripped the extra particles off the top piece of tape b. positive, because it was attracted to the plastic...
- C Chemistry Jan has a cat that is about to have kittens. She wonders how much they will look like their mother. Which of the following best explains the physical resemblance that kittens...
- C Chemistry Select the correct answer from each drop-down menu. How are symbols used in an architectural drawing? symbols show the picture of the finished object from the front, while entire...
- C Chemistry Calculate the mass percent by volume of 330.1 g of glucose (C₆H₁₂O₆, MM = 180.2 g/mol) in 325 mL of solution....
- C Chemistry Predict the two most likely mechanisms for the reaction of 1-iodohexane with potassium tert-butoxide. A) E1 and SN1 B) SN1 and SN2 C) SN2 and E2 D) E1 and E2...
- C Chemistry Which one of the following best represents the predicted approximate chemical shift and coupling for the hydrogen(s) indicated with the arrow? a) 5.40 ppm, doublet b) 3.00 ppm,...
- C Chemistry For the dissolution of LiCl in water, ΔHsoln=−37kJ/mol. Which term would you expect to be the largest negative number: ΔHsolvent, ΔHsolute, or ΔHmix? a. ΔHsolvent b. ΔHsolute...
- E English Which electron configuration of the 4f energy sublevel is the most stable? A-4f13 B-4f14 C-4f D-4f7...
- M Mathematics The binomial expansion 625x^4 – 7,500x^3 + 33,750x^2 – 67,500x + 50,625 can be expressed as (ax + b)^4. What is the value of b? –15–5 5 15...
- M Mathematics Juan is purchasing a brand new Ford Mustang that costs $38,500. The car depreciates 12% annually. Which equation below can be used to determine the value of the car 5 years...

Ответ:
Answer : The equilibrium constant kc is 4.76 x 10⁻³
Explanation :
The given equilibrium reaction is
Step 1 : Set up ICE table
Let us set up an ICE table for this reaction .
The initial concentration of SO₃ is
The initial concentrations of products are 0.
Let us assume x is the change .
Please refer the attached picture.
Step 2 : Use the given value to find x
From the ICE table, we can see that at equilibrium, concentration of O₂ is x
But we have been given that , at equilibrium we have 0.190 mol of O₂ .
Let us convert this to concentration unit.
Concentration of O₂ at equilibrium =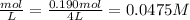
But concentration of O₂ from the ICE table is x.
Therefore we have x = 0.0475 M
Step 3 : Using x , find equilibrium concentrations
Using this value, let us write the equilibrium concentrations of the given species.
[SO₃]eq = 0.185 M - 2x = 0.185 - 2(0.0475) = 0.09 M
[SO₂]eq = 2x = 0.095 M
[O₂]eq = x = 0.0475 M
Step 4 : Set up equation for kc and solve it
The equilibrium constant kc is calculated as,
Let us plug in the above equilibrium values.
The equilibrium constant kc is 4.76 x 10⁻³
Ответ:
isolation
Explanation: