kromaug1517
01.07.2021 •
Chemistry
Calculate the Experimental Molar Volume in L/mol of the Hydrogen gas, H2, if the volume of H2 at STP is 52.8 mL and the mass of Magnesium metal, Mg, used in the experiment is 0.055 g.
Solved
Show answers
More tips
- S Style and Beauty Ultimate Guide on How to Care for Suede Shoes...
- S Sport Running: How to Do It Right?...
- F Food and Cooking How to Cook Spaghetti Right – Secrets and Tips...
- P Philosophy Personal attitude towards Confession: how to prepare and undergo the procedure correctly?...
- H Health and Medicine Flu: How to Recognize It by the First Symptoms?...
- F Food and Cooking How to Sober Up Quickly? Important Facts and Tips...
- H Health and Medicine How to Properly Take a Blood Sugar Test?...
- H Health and Medicine Simple and Effective: How to Get Rid of Cracked Heels...
- O Other How to Choose the Best Answer to Your Question on The Grand Question ?...
- L Leisure and Entertainment History of International Women s Day: When Did the Celebration of March 8th Begin?...
Answers on questions: Chemistry
- B Business Alpha Industries is considering a project with an initial cost of $8.8 million. The project will produce cash inflows of $1.68 million per year for 8 years. The project...
- M Mathematics Construct the matrix diagram for each of the following models. breading systme: coorperatively breeding bird species are those in which some individuals forgo their own...
- S Social Studies Rita, a follower of a certain religion, posts a podcast on the sabbath! website in which she insists that congress base all federal law on her religious principles. under...
- E English Read the excerpt from Sinners in the Hands of an Angry God. We find it easy to tread on and crush a worm that we see crawling on the earth; so it is easy for us to cut...
- H History Which of the following benefitted MOST from the Suez Canal?...

Ответ:
The Experimental Molar Volume in L/mol of the Hydrogen gas=23.36L/mol
Explanation:
We are given that
Volume of H2 at STP=52.8mL
Mass of magnesium metal ,M(Mg)=0.055g
We have to find the Experimental Molar Volume in L/mol of the Hydrogen gas.
Molar mass of Mg=24.305 g/mol
Number of moles=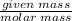
Using the formula
Number of moles of Mg= moles
moles
Number of moles of Mg=0.00226moles
Number of moles of Mg=Number of moles of H2
Number of moles of H2=0.00226moles
Molar volume of Hydrogen gas (H2)=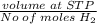
Molar volume of Hydrogen gas (H2)=
Molar volume of Hydrogen gas (H2)=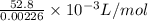
Molar volume of Hydrogen gas (H2)=23.36L/mol
Hence, the Experimental Molar Volume in L/mol of the Hydrogen gas=23.36L/mol
Ответ:
I dont know if this is the right subject but i got an A+ on it. It is Life science edgenuity This is the process part of it
Explanation:
First, I observed all of the arthropods that I would be using in my dichotomous key, Fire Ant, Bee, Butterfly, Dragonfly, Ladybug, Scorpion, Spider, and Wasp. I recorded my observations on a characteristics table that included Legs, Wings, Stinger, Antennae, and Claws.
Next, I used my observations to create my first question, “Does the arthropod have a stinger?).
Then, I Looked over my notes and found the arthropds that have stingers, Fire Ant, Bee, Scorpion, and Wasp, and sorted them into a Yes group. Also I found the arthropods in my notes that didn’t have stingers, Butterfly, Dragonfly, Ladybug, and Spider, and put them into a No group.
After That, I Chose a new question coming off of the Yes group, “Does the arthropod have 6 legs?”. I sorted those the arthropods that did have 6 legs into a Yes group, Fire Ant, Bee, and Wasp.
Then, I did the same for The arthropods who didn’t have 6 legs and the only one was Scorpion, so I wrote scorpion in the No group and that Branch was done.
Next, I did another question for my very first No group, “Does the arthropod have antennae?” and I sorted the arthropods into the yes and no branches. No group included, Dragonfly and Spider. Yes group included, Butterfly and Ladybug.
Later, I did a question for the yes group coming off of the 6 legs question, “Does the arthropod have wings?”. The yes group included, Bee and Wasp. The no group included, the Fire Ant only so I wrote down Fire Ant and that branch was concluded.
Then, I did a question for the no group coming off of the antennae question, “Does the arthropod hve 8 legs?” and my yes group included spider only so that branch was done. My no group included Dragonfly so that branch was concluded aswell.
After That, I wrote a question for the yes group coming off of the wings question, “Does the arthropod have 2 wings?”. My Yes group concluded wit only the Wasp left so I had finished that branch. I finished my no group with a Bee.
Lastly, I did my last question of the key coming off of the Yes group of the antennae question, “Does the arthropod have 4 wings?”. My no group included a Ladybug and my yes group included a Butterfly.
In conclusion, My dichotomous key was finished with 8 athropods in each branch alone and now you can use the dichotomous to observe a Fire Ant, Bee, Wasp, Butterfly, Dragonfly, Ladybug, Spider, and Scorpion.
Please mark me brainliest :) :) :) :) :) :)