Calculate the volume of hydrogen gas that is produced by the reaction of 0.52 grams of solid magnesium with excess hydrochloride acid (HCl). This is a single replacement reaction.
Solved
Show answers
More tips
- F Food and Cooking Apple Cider Vinegar: The Ultimate Health and Beauty Solution...
- C Computers and Internet Е-head: How it Simplifies Life for Users?...
- F Family and Home How to Choose the Best Diapers for Your Baby?...
- F Family and Home Parquet or laminate, which is better?...
- L Leisure and Entertainment How to Properly Wind Fishing Line onto a Reel?...
- L Leisure and Entertainment How to Make a Paper Boat in Simple Steps...
- T Travel and tourism Maldives Adventures: What is the Best Season to Visit the Luxurious Beaches?...
- H Health and Medicine Kinesiology: What is it and How Does it Work?...
- O Other How to Choose the Best Answer to Your Question on The Grand Question ?...
- L Leisure and Entertainment History of International Women s Day: When Did the Celebration of March 8th Begin?...
Answers on questions: Chemistry
- C Chemistry Someone help. trying to past this test but keep failing on this question. Atoms may emit light energy when a. protons move to a higher energy level b. protons move to a lower energy...
- C Chemistry NEED HELP ASAP: define and explain the trend of atomic radius and name an element that is larger than carbon......
- C Chemistry 2AgCl + H2 → 2Ag + 2HCI...
- C Chemistry El CH₂ + 202 + CO₂2 H₂O reacción química...
- C Chemistry What total volume of ozone measured at a pressure of 24.5 mmHg and a temperature of 232 K can be destroyed when all of the chlorine from 15.5 g of CF3Cl goes through 10 cycles...
- C Chemistry Two moles of acetyl chloride was mixed with two moles of dimethylamine. After the reaction is complete, what species can be found in the mixture...
- C Chemistry What products will most likely form when a carboxylic acid reacts with an amine at high temperatures, or in the presence of a catalyst...
- C Chemistry If KI K I has a solubility of 140. G g in 100. G g of H2O H 2 O at 20 ∘C ∘ C , how many grams of water are needed to prepare a saturated solution containing 75.0 g g of KI K I...
- C Chemistry What molecules can be broken down to one Another ?...
- C Chemistry The best description of atoms or molecules that have a positive or negative charge and are dissolved in body fluids is: Select one: O a. Amino acids b. Acids Base d. Sugars e....

Ответ:
In this given case, he volume of hydrogen gas produced = 0.03969 L
Given:
0.52 grams - solid magnesium
Solution:
moles of Mg
=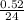
= 0.216 x 10⁻³
Reaction -
Mg + 2HCl -----------------> MgCl2 + H2
1mol 1mol
0.216 x 10⁻³ 0.216 x 10⁻³ mol
moles of H2
= 0.216 x 10⁻³
T = 273 K
P =1 atm
From idea gas P V = n R T
Placing given and known values in ideal gas
1 x V = 0.216 x 10⁻³ x 0.0821 x 273
V = 0.048 L
Thus, the volume of hydrogen gas produced = 0.03969 L
Learn more:
link
Ответ:
0.493L
Explanation:
Let us write a balanced equation for the reaction. This is illustrated below:
Mg + 2HCl —> MgCl2 + H2
Next, let us convert 0.52g of Mg to mole. This is illustrated below:
Molar Mass of Mg = 24g/mol
Mass of Mg = 0.52g
Number of mole of Mg = Mass /Molar Mass = 0.52/24 = 0.022mole
From the equation,
1mole of Mg produced 1mole of H2.
Therefore, 0.022mole of Mg will always produce 0.022mole of H2.
1mole of H2 occupies 22.4L at stp.
Therefore, 0.022mole of H2 will occupy = 0.022 x 22.4 = 0.493L
Ответ:
Al escribirla, he tenido cuatro objetivos presentes. En primer lugar, re- trasé su redacción y publicación hasta que estuve bastante seguro de no in-.
270 páginas