Can some one me with science . i am having a difficult time .

Solved
Show answers
More tips
- F Food and Cooking Effective Methods to Organize Videos in your iPad According to Content...
- F Family and Home Parquet or laminate, which is better?...
- L Leisure and Entertainment How to Properly Wind Fishing Line onto a Reel?...
- L Leisure and Entertainment How to Make a Paper Boat in Simple Steps...
- T Travel and tourism Maldives Adventures: What is the Best Season to Visit the Luxurious Beaches?...
- H Health and Medicine Kinesiology: What is it and How Does it Work?...
- O Other How to Choose the Best Answer to Your Question on The Grand Question ?...
- L Leisure and Entertainment History of International Women s Day: When Did the Celebration of March 8th Begin?...
- S Style and Beauty Intimate Haircut: The Reasons, Popularity, and Risks...
- A Art and Culture When Will Eurovision 2011 Take Place?...
Answers on questions: Chemistry
- C Chemistry What is the electron geometry of CO2 ?...
- C Chemistry Helpppp ppleaseeee ill mark you as brainlister...
- C Chemistry What type of reaction is this? If 40.0 grams of magnesium is reacted with an excess of nitric acid, HNO3, how many grams of hydrogen gas will be produced?...
- C Chemistry Why do you think earthquakes appear in the places they do?...
- C Chemistry Use the periodic table to determine the amount of copper and aluminum nitrate formed in the reaction. Type the correct answer in the box. Express your answer to the correct...
- C Chemistry The pressure in a certain tire is 109 kPa, what is the pressure of the air in the tire in psi?...
- C Chemistry What is KOH + HCIO CL + FeH + Cl...
- C Chemistry Astudent neutralized 16.4 milliliters of hcl by adding 12.7 milliliters of 0.620 m koh. what was the molarity of the hcl acid? (1) 0.168 m (3) 0.620 m (2) 0.480 m (4)...
- C Chemistry Metamorphic rocks that don’t have layers true or false...
- C Chemistry Muscles convert chemical energy into...

Ответ:
Kilometer - km - 1,000
Hectameter - hm - 100
Decameter - dam - 10
Millimeter - mm - 0.001
The table provides clear prefix rules.
The prefixes can be used for most (if not all) units such as grams (kg,mg...).
Ответ:
The partial pressure of nitrogen gas is 405.76 mmHg and that of hydrogen gas is 257.24 mmHg
Explanation:
To calculate the number of moles, we use the equation:

For nitrogen gas:Given mass of nitrogen gas = 3.46 g
Molar mass of nitrogen gas = 28 g/mol
Putting values in above equation, we get:
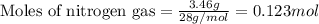
For hydrogen gas:Given mass of hydrogen gas = 0.156 g
Molar mass of hydrogen gas = 2 g/mol
Putting values in above equation, we get:
Mole fraction of a gas is calculated by using the formula:
 ......(1)
......(1)
For nitrogen gas:Putting values in equation 1, we get:

For hydrogen gas:Putting values in equation 1, we get:
The partial pressure of a gas is given by Raoult's law, which is:
where,
 = mole fraction of substance A
= mole fraction of substance A
For nitrogen gas:
For hydrogen gas:Hence, the partial pressure of nitrogen gas is 405.76 mmHg and that of hydrogen gas is 257.24 mmHg