valleriieZ7002
07.07.2020 •
Chemistry
Carbon monoxide (CO) reacts with hydrogen (H2) to form methane (CH4) and water (H20).
CO(g) + 3H2(g) + CH4(g)+H20(9)
The reaction is at equilibrium at 1,000 K. The equilibrium constant of the reaction is 3.90. At equilibrium, the concentrations are as
follows.
[CO] = 0.30 M
[H2] = 0.10 M
[H20] = 0.020 M
What is the equilibrium concentration of CH, expressed in scientific notation?
0.0059
5.9 x 10-2
0.059
5.9 x 102
Solved
Show answers
More tips
- F Food and Cooking 10 Tips for Proper Sushi Consumption...
- H Health and Medicine Want to Lose Weight? Gain Muscle without Damaging Your Health!...
- F Family and Home Parquet or laminate, which is better?...
- L Leisure and Entertainment How to Properly Wind Fishing Line onto a Reel?...
- L Leisure and Entertainment How to Make a Paper Boat in Simple Steps...
- T Travel and tourism Maldives Adventures: What is the Best Season to Visit the Luxurious Beaches?...
- H Health and Medicine Kinesiology: What is it and How Does it Work?...
- O Other How to Choose the Best Answer to Your Question on The Grand Question ?...
- L Leisure and Entertainment History of International Women s Day: When Did the Celebration of March 8th Begin?...
- S Style and Beauty Intimate Haircut: The Reasons, Popularity, and Risks...
Answers on questions: Chemistry
- C Chemistry A can of butane with a pressure of 2 atm at 283k has its pressure increased to a new pressure of 5 atm. What is the temperature of the new container?...
- C Chemistry the largest temperature decline during a 1 day (24 hours) time period was 56 degrees Celsius in Browning, Montana. Express this as degrees Fahrenheit per minute...
- C Chemistry What is the most soluble salt of the following set? what is the most soluble salt of the following set? (a) sn(oh)2 with ksp = 1.6 × 10-19 (b) al(oh)3 with ksp = 1.9 × 10-33...
- C Chemistry Consider a process in which 100 kj of energy is transferred reversibly and isothermally as heat to a large block of copper. calculate the change in entropy of the block if the...
- C Chemistry Given two half reactions as follows: a2+ → 2 a2+ + 3 e− 4 e− + b → b4− what would you multiply each half-reaction by, to cancel out the electrons?...
- C Chemistry For each of the following pairs of polymers, plot and label schematic stress–strain curves on the same graph [i.e., make separate plots for parts (a) to (c)]. (a) polyisoprene...
- C Chemistry Aprecipitation reaction is caused by mixing 100. ml of 0.25 m k2cr2o7 solution with 100. ml of 0.25 m pb(no3)2 solution. when the precipitate forms, it is filtered from the mixture.how...
- C Chemistry The most useful ore of aluminum is bauxite, in which al is present as hydrated oxides, al2o3 * xh2o. a) the number of kilowatt-hours of electricity required to produce 3.00kg...
- C Chemistry Identify the chemical formula for calcium phosphate. A) Ca2(PO4)3 B) Ca3(PO3)2 C) Ca3(PO4)2 D) CaPO4...
- C Chemistry What are the answers? pls need help! letters B-D...

Ответ:
The equilibrium concentration of , expressed in scientific notation is
, expressed in scientific notation is 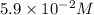
Explanation:
Equilibrium constant is the ratio of the concentration of products to the concentration of reactants each term raised to its stochiometric coefficients.
The given balanced equilibrium reaction is,
At eqm. conc. (0.30) M (0.10) M (x) M (0.020) M
The expression for equilibrium constant for this reaction will be,
Now put all the given values in this expression, we get :
By solving the term 'x', we get :
x = 0.059 M=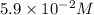
Thus, the concentrations of at equilibrium is :
at equilibrium is :
Concentration of = (x) M =
= (x) M = 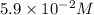
The equilibrium concentration of , expressed in scientific notation is
, expressed in scientific notation is 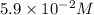
Ответ:
Take in carbon dioxide, reduces water loss
Explanation: