19dansiste
26.12.2019 •
Chemistry
Find the protons, electrons and neutrons for atom with a mass of 3 an an atomic number of 1. i think the atom is hydrogen, but i don't have a clue about the mass. !
Solved
Show answers
More tips
- S Style and Beauty How to Make Your Lips Fuller? Ideas and Tips for Beautiful Lips...
- S Style and Beauty How are artificial nails removed?...
- F Family and Home How to Sew Curtain Tapes: Best Tips from Professionals...
- H Horoscopes, Magic, Divination How to Cast a Love Spell on a Guy? Guide for Guys...
- F Family and Home How to Properly Use a Water Level?...
- L Legal consultation What Documents Are Required for a Russian Passport?...
- H Health and Medicine How to Treat Styes: Causes, Symptoms, and Home Remedies...
- F Family and Home Protect Your Home or Apartment from Pesky Ants...
- T Travel and tourism Lost in the Catacombs: What to Do?...
- C Computers and Internet Make Easy Accessible Screenshots on iPad in Just a Few Minutes...
Answers on questions: Chemistry
- B Business Develop a training budget assuming that the trainer is paid $140 per hour, that 8 hours are needed to develop training materials, and that the 5 workers earn $18...
- S Spanish Please help very simple question but im not good at spanish....
- M Mathematics a person has 80 followers on social media. The number of followers tripled each year. How many followers will she have after 4 years. Explain your answer...
- S Social Studies Stoneville pedigreed seed company produces genetically modified cotton seeds. what are some advantages to using genetically modified crops?...

Ответ:
protons=1
electrons=1
neutrons=0
Explanation:
protons and electrons are equal in their natural state when it goes under any reaction the protons of that element remain the same but the number of the element changes.
Ответ:
8.771 grams of AgCl2 is formed from the reaction of 10.4 g of silver nitrate reacts with 15.0 g of barium chloride.
Explanation:
The balanced equation for the reaction between silver nitrate and barium chloride is shown as:
2AgNO3 + BaCl2⇒ 2AgCl
From the reaction and the quantity of reactants given the limiting reagent could be calculated.
For limiting reagent we calculate the amount of AgCl formed from the quantity of reactants.
15 gram of BaCl2 gives
number of moles=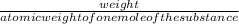
number of moles =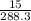
= 0.052 moles of BaCl2 is used
1 mole of BaCl2 yielded 2 moles of AgCl
0.052 moles of BaCl2 will yield x moles
= 0.104 moles of AgCl is formed.
10.4 grams of siver nitrate gives
number of moles=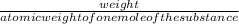
number of moles =
= 0.0612 moles of AgNO3
so, 2 moles of AgNO3 forms 2 mole of AgCl
0.0612 moles will form x moles of AgCl
0.0612 moles of Agcl will be formed
It is concluded that the lowest number of moles of AgCl is produced was from AgNO3 so AgNO3 is a limiting reagent in the reaction.
Thus, the least moles of AgCl formed will be converted to grams
0.0612 x 143.32
= 8.771 grams