Help me out ! What’s the answer
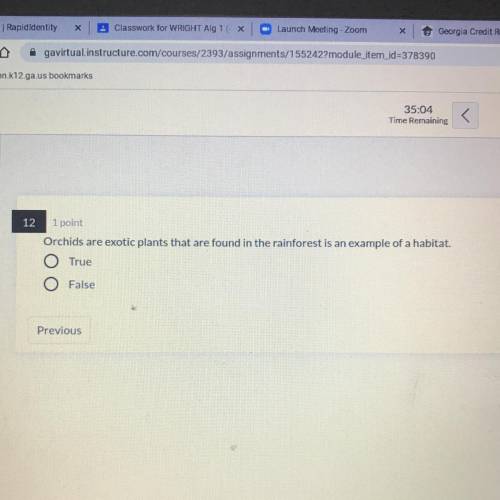
Solved
Show answers
More tips
- L Leisure and Entertainment How to Make a Paper Boat in Simple Steps...
- T Travel and tourism Maldives Adventures: What is the Best Season to Visit the Luxurious Beaches?...
- H Health and Medicine Kinesiology: What is it and How Does it Work?...
- O Other How to Choose the Best Answer to Your Question on The Grand Question ?...
- L Leisure and Entertainment History of International Women s Day: When Did the Celebration of March 8th Begin?...
- S Style and Beauty Intimate Haircut: The Reasons, Popularity, and Risks...
- A Art and Culture When Will Eurovision 2011 Take Place?...
- S Style and Beauty How to Choose the Perfect Hair Straightener?...
- F Family and Home Why Having Pets at Home is Good for Your Health...
- H Health and Medicine How to perform artificial respiration?...
Answers on questions: Chemistry
- B Business The marginal rate of technical substitution always equals A. minus the ratio of the marginal products of inputs. B. the slope of the total product curve. C. the change in output...
- P Physics Definition of artficial and natural radioactivity...
- H Health 2 What is aerobic exercise? 3. What is anaerobic exercise? 4. Give 2 examples of aerobic exercise. 5. Give 2 examples of anaerobic exercise....

Ответ:
true
Explanation:
Ответ:
55.0 g
Explanation:
Step 1: Write the balanced equation for the production of oxygen
2 CO₂(g)⇒ 2 CO(g) + O₂(g)
Step 2: Calculate the mass of oxygen produced over a 2 hour period
The MOXIE produces 10.0 g of oxygen per hour.
2 h × 10.0 g/1 h = 20.0 g
Step 3: Calculate the moles corresponding to 20.0 g of O₂
The molar mass of O₂ is 32.00 g/mol.
20.0 g × 1 mol/32.00 g = 0.625 mol
Step 4: Calculate the number of moles of CO₂ needed to produce 0.625 moles of O₂
The molar ratio of CO₂ to O₂ is 2:1. The moles of CO₂ needed are 2/1 × 0.625 mol = 1.25 mol
Step 5: Calculate the mass corresponding to 1.25 moles of CO₂
The molar mass of CO₂ is 44.01 g/mol.
1.25 mol × 44.01 g/mol = 55.0 g