How do you predict the charge of the atom using Bohr’s model? Please explain using an example.
Solved
Show answers
More tips
- S Style and Beauty Ultimate Guide on How to Care for Suede Shoes...
- S Sport Running: How to Do It Right?...
- F Food and Cooking How to Cook Spaghetti Right – Secrets and Tips...
- P Philosophy Personal attitude towards Confession: how to prepare and undergo the procedure correctly?...
- H Health and Medicine Flu: How to Recognize It by the First Symptoms?...
- F Food and Cooking How to Sober Up Quickly? Important Facts and Tips...
- H Health and Medicine How to Properly Take a Blood Sugar Test?...
- H Health and Medicine Simple and Effective: How to Get Rid of Cracked Heels...
- O Other How to Choose the Best Answer to Your Question on The Grand Question ?...
- L Leisure and Entertainment History of International Women s Day: When Did the Celebration of March 8th Begin?...
Answers on questions: Chemistry
- C Chemistry BioAgent vs Green Zombie Toxin...
- C Chemistry Which of the following substances would be most likely to float in water (density 1.0 g/mL)? A. A substance with a density of 10 kg/L B. A substance with a density of 2 mg/cL C. A...
- C Chemistry When a chlorate decomposes what is produced? none of these O Oxygen gas O Water Carbon dioxide Sulfur trioxide...
- C Chemistry 5. A sample of pure acetyl salicylic acid had a mass of 0.268 grams. It was dissolved in NaOH and diluted to 500.00mL in a volumetric flask. a. What is the concentration of aspirin...
- C Chemistry How many carbon atoms are in 2.0 moles of CH4 ?...
- C Chemistry In thin layer chromotagraphy, one should place as much of the sample to be separated as possible onto the slide. True or false...
- C Chemistry What is the unscrambled word in science for mueeorgvroaste...
- C Chemistry Describe 2 ways you can conserve energy in your home - and do not just type reduce, reuse, recycle! describe 2 actual things you can do to save energy....
- C Chemistry What is the most threatening, long term,aspect of nuclear fallout? a) intense light b)explosive power c)thermal exposure d)radioactive contamination...
- C Chemistry If i have 7.7 moles of gas at a pressure of 0.0990 atm and at a temperature of 256 k, what is the volume of the container that the gas is in?...

Ответ:
The charge of the atom using Bohr’s model is present below.
Explanation:
According to Bohr's model, there is no charge on the atom if the number of proton and electron are same or in other words, if an atom does not gain or lose electron so there is no charge on it. If the atom loses electron, a positive charge appears on it due to increase occurs in the number of proton, while if the atom receives the electron, negative charge will appear on it due to increase in number of electron. For example, when sodium react with chlorine, it loses electron, a positive charge appears on it while when chlorine receives the electron, negative charge appears on it making sodium chloride.
Ответ:
pH of buffer before addition of Strong acid = 4.71
pH of buffer after addition of Strong acid = 2
Explanation:
According to Henderson's equation
⇒pH = pKa +㏒![\frac{[salt]}{[Acid]}](/tpl/images/0555/8558/c4480.png)
⇒pH = 4.74 + log(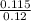 )
)
⇒pH = 4.74 - 0.03
⇒pH = 4.71
⇒[H⁺] = 1.9 x 10⁻⁵
pH of this buffer after 0.01 mole of strong acid addition
⇒pH = - log[H⁺]
⇒pH = - log(1.9 x 10⁻⁵ + 0.01)
⇒pH = 2