How long would it take to produce enough aluminum to make a case (24 cans) of aluminum soft drink cans if each can used 3 g of aluminum, a current of 5.00 x 104 A was employed, and the current efficiency was 93.9 %?
Solved
Show answers
More tips
- S Sport How to Pump Your Chest Muscle? Secrets of Training...
- H Horoscopes, Magic, Divination Where Did Tarot Cards Come From?...
- S Style and Beauty How to Make Your Lips Fuller? Ideas and Tips for Beautiful Lips...
- S Style and Beauty How are artificial nails removed?...
- F Family and Home How to Sew Curtain Tapes: Best Tips from Professionals...
- H Horoscopes, Magic, Divination How to Cast a Love Spell on a Guy? Guide for Guys...
- F Family and Home How to Properly Use a Water Level?...
- L Legal consultation What Documents Are Required for a Russian Passport?...
- H Health and Medicine How to Treat Styes: Causes, Symptoms, and Home Remedies...
- F Family and Home Protect Your Home or Apartment from Pesky Ants...
Answers on questions: Chemistry
- C Chemistry Which order shows the levels of organization from largest to smallest? ОООО organism, organ system, cell, organ, tissue organism, tissue, organ system, organ, cell organism,...
- C Chemistry What are zebra mussels?...
- C Chemistry What is chemistry for people who hate other people?...
- C Chemistry Fluorine-19 is an isotope of fluorine. it has a different number of electrons (a) protons (b) neutrons (c) electron shells (d)...
- C Chemistry Uniformitarianism states that the geologic processes occurring in the past are the same processes that are occurring today. Processes we see happening on Earth today have...
- C Chemistry Please help I have a test...
- C Chemistry En los gases la fuerza de repulsión intermolecular, es mayor que la fuerza de atracción es verdadero o falso?...
- C Chemistry Which statements describe or apply the principal of conservation of energy? Select all that apply. * A. Energy cannot be created or destroyed; it can only be transferred...
- C Chemistry A .875 L capsule with 2.58 mol of toxic gas at constant temperature happens to have a opened valve allowing the toxic gas to enter a 52.0 L shed with lethal Deadly Nightshade...
- C Chemistry 30: Unit D Assignment Booklet D1 39. An example of an exothermic fusion reaction is H+ H+ H+ n. The energy released from this reaction is due to a small change in mass...

Ответ:
Answer : It takes time to produce 3 g of aluminium is, 165 seconds.
Explanation :
As we are given that the mass of aluminium is, 3 grams. Now we have to calculate the total mass of aluminum.
Total mass of aluminum = 3g × 24 = 72 g
Now we have to calculate the moles of aluminum.
Molar mass of Al = 27 g/mol
As, 1 mole of Al has 3 Faradays
So, 2.67 mole of Al has = Faradays
Faradays
and,
Charge =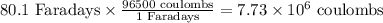
Current =
Now we have to calculate the time.
Hence, it takes time to produce 3 g of aluminium is, 165 seconds.
Ответ:
Hope this helps!