dnprops1544
27.01.2020 •
Chemistry
If 75.0 g silderite ore (feco3) is heated with an excess of oxygen, 45.0 g of ferric oxide (fe2o3) is produced. 4feco3(s) + o2(g) → 2fe2o3(s) + 4co2(g) what is the percent yield of this reaction?
Solved
Show answers
More tips
- H Health and Medicine Want to Lose Weight? Gain Muscle without Damaging Your Health!...
- F Family and Home Parquet or laminate, which is better?...
- L Leisure and Entertainment How to Properly Wind Fishing Line onto a Reel?...
- L Leisure and Entertainment How to Make a Paper Boat in Simple Steps...
- T Travel and tourism Maldives Adventures: What is the Best Season to Visit the Luxurious Beaches?...
- H Health and Medicine Kinesiology: What is it and How Does it Work?...
- O Other How to Choose the Best Answer to Your Question on The Grand Question ?...
- L Leisure and Entertainment History of International Women s Day: When Did the Celebration of March 8th Begin?...
- S Style and Beauty Intimate Haircut: The Reasons, Popularity, and Risks...
- A Art and Culture When Will Eurovision 2011 Take Place?...
Answers on questions: Chemistry
- C Chemistry The upper quartile of a set of data is a number that a. separates the bottom half of the data set from the upper half b. finding the mean of the data set remaining...
- C Chemistry How does the atmospheric pressure vary with height?...
- C Chemistry In the reaction, 210 kJ of heat energy is used to form 3.0 moles of hydrogen. Calculate how much heat energy is needed to make 1000 kg of hydrogen....
- C Chemistry 5.) Could the number 45.72 be an atomic number, average atomic mass, or both?...
- C Chemistry Does the biosphere have clear boundaries...
- C Chemistry Calculate the residence time of sodium. fill in the blanks. use t= m/f mass (m) flow rate (f) ton ton/year sodium 2.8 x 10^13 3.5 x 10^5 zach is investigating the...
- C Chemistry Which of the following use of chemicals is correct? urgent questiona. window cleaner is used to clean a food contact surfaceb. chemicals are used at double the recommended...
- C Chemistry To cool foods quickly, place thema. in the refrigirator in deep containers.b. in refrigirator in large pots with tight fitting lids.c. in refrigirator after cooling...
- C Chemistry What is the pH of 1 x 10 M HCl solution?...
- C Chemistry 28.The chemical formula of a compound indicates the source of the elements in the compound. b. how elements are joined in the compound. the alchemy symbols for the...

Ответ:
Answer : The percent yield of the reaction is, 87.0 %
Solution : Given,
Mass of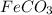 = 75.0 g
= 75.0 g
Molar mass of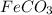 = 115.85 g/mole
= 115.85 g/mole
Molar mass of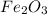 = 159.69 g/mole
= 159.69 g/mole
First we have to calculate the moles of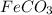 .
.
Now we have to calculate the moles of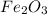
The balanced chemical reaction is,
From the balanced reaction we conclude that
As, 4 mole of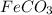 react to give 2 mole of
react to give 2 mole of 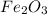
So, 0.647 moles of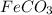 react to give
react to give 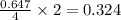 moles of
moles of 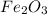
Now we have to calculate the mass of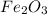
Theoretical yield of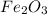 = 51.7 g
= 51.7 g
Experimental yield of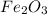 = 45.0 g
= 45.0 g
Now we have to calculate the percent yield of the reaction.
Therefore, the percent yield of the reaction is, 87.0 %
Ответ:
you would need to add or mabye multiply the divide
Explanation: