If a 25 kg object is moving at a velocity of 5 m/s
Solved
Show answers
More tips
- H Health and Medicine How to Increase Hemoglobin in the Blood...
- A Animals and plants How to Store Carrots: Tips for Homeowners...
- L Legal consultation Juvenile Justice: Who Needs It?...
- F Family and Home How to Choose the Best Diapers for Your Baby?...
- F Family and Home Parquet or laminate, which is better?...
- L Leisure and Entertainment How to Properly Wind Fishing Line onto a Reel?...
- L Leisure and Entertainment How to Make a Paper Boat in Simple Steps...
- T Travel and tourism Maldives Adventures: What is the Best Season to Visit the Luxurious Beaches?...
- H Health and Medicine Kinesiology: What is it and How Does it Work?...
- O Other How to Choose the Best Answer to Your Question on The Grand Question ?...
Answers on questions: Chemistry
- C Chemistry Someone please help with this...
- C Chemistry How many nonbonding pairs of electrons are there around the central sulfur atom in H2S? Given : S atomic number 16 *...
- C Chemistry Methyl violet is an indicator that changes color over a range from ph=0.0 to ph=1.6. what is ka of methyl violet?...
- C Chemistry All of the following statements about the atmosphere are true except: a. the biosphere includes all living organisms on earth s surface. b.like biology the name for the...
- C Chemistry How does hydrogen peroxide bleach hair? a. the h2o2 molecules break the double bonds in pigment molecules. b. melanin changes h2o2 molecules to double bonds c. the h2o2...
- C Chemistry Acetone [(CH3)2C=O], a simple ketone is commonly used to make products like nail polish remover and paint remover. Draw a picture showing the orbitals involved in the...
- C Chemistry Aro stated that when someone hits a golf ball with a club, the amount of energy the ball has changes, the amount of energy that the club has changes, and the total energy...
- C Chemistry Given that 21 kg of magnesium carbonate is heated, calculate the mass of magnesium oxide formed?...
- M Mathematics Chords bc and de intersect at point f as shown what is the length of de...
- M Mathematics Which statement is true about the songs played during the one-hour interval? Most of the songs were between 3 minutes and 3.8 minutes long. Most of the songs were 3.4...

Ответ:
The empirical and molecular formula for the given organic compound is CH and and it is not an alkane.
and it is not an alkane.
Explanation:
The chemical equation for the combustion of hydrocarbon having carbon and hydrogen follows:
where, 'x' and 'y' are the subscripts of Carbon, hydrogen and oxygen respectively.
We are given:
Mass of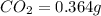
Mass of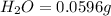
We know that:
Molar mass of carbon dioxide = 44 g/mol
Molar mass of water = 18 g/mol
For calculating the mass of carbon:
In 44 g of carbon dioxide, 12 g of carbon is contained.
So, in 0.364 g of carbon dioxide, of carbon will be contained.
of carbon will be contained.
For calculating the mass of hydrogen:
In 18 g of water, 2 g of hydrogen is contained.
So, in 0.0596 g of water,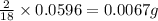 of hydrogen will be contained.
of hydrogen will be contained.
To formulate the empirical formula, we need to follow some steps:
Step 1: Converting the given masses into moles.Moles of Carbon =
Moles of Hydrogen =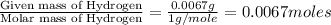 ]
]
Step 2: Calculating the mole ratio of the given elements.For the mole ratio, we divide each value of the moles by the smallest number of moles calculated which is 0.0067 moles.
For Carbon =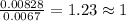
For Hydrogen =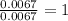
Step 3: Taking the mole ratio as their subscripts.The ratio of C : H = 1 : 1
The empirical formula for the given compound is CH
For determining the molecular formula, we need to determine the valency which is multiplied by each element to get the molecular formula.
The equation used to calculate the valency is:
We are given:
Mass of molecular formula = 128.2 g/mol
Mass of empirical formula = 13 g/mol
Putting values in above equation, we get:
Multiplying this valency by the subscript of every element of empirical formula, we get:
The general formula of an alkane is , where n = any natural number
, where n = any natural number
Here, n = 10 and it does not satisfy being an alkane
Hence, the empirical and molecular formula for the given organic compound is CH and and it is not an alkane.
and it is not an alkane.