ttangelique
22.10.2019 •
Chemistry
Percentage compostion of carbon in caco3
Solved
Show answers
More tips
- B Business and Finance How to Create a Business Plan? Your Ultimate Guide...
- F Family and Home Parquet or laminate, which is better?...
- L Leisure and Entertainment How to Properly Wind Fishing Line onto a Reel?...
- L Leisure and Entertainment How to Make a Paper Boat in Simple Steps...
- T Travel and tourism Maldives Adventures: What is the Best Season to Visit the Luxurious Beaches?...
- H Health and Medicine Kinesiology: What is it and How Does it Work?...
- O Other How to Choose the Best Answer to Your Question on The Grand Question ?...
- L Leisure and Entertainment History of International Women s Day: When Did the Celebration of March 8th Begin?...
- S Style and Beauty Intimate Haircut: The Reasons, Popularity, and Risks...
- A Art and Culture When Will Eurovision 2011 Take Place?...
Answers on questions: Chemistry
- M Mathematics Interpret units in formulas Problem Consider the formula W=FdW=FdW, equals, F, d, where WWW represents work, FFF represents force and has units of Newtons \left( \dfrac{\text{kg}\cdot\text{m}}{\text{s}^2}\right)(...
- E English What is something you ve learned from The Crucible so far that you can apply to your life? 3 complete sentences for full points;)....
- B Business Laws issued by a government agency are laws. A.RegulatoryB.CustomC.statutoryD.Administrative...
- C Computers and Technology Plz help, will guve brainliest to best answer (if i can)...

Ответ:
C = 12.01 amu
O = 16 * 3 = 48 amu
40.08 + 12.01 + 48 = 100.09 total amu
12.01 / 100.09 = 0.1199 * 100 = about 12% C
To find percent composition carbon you do
12.01 amu divided by the total amu of compound times 100..
amu is just the atomic mass number of the element/compound
Ответ:
The ratio of mass of sulfur atom to mass of oxygen atom in sulfur dioxide is 1: 1.
Explanation:
Law of constant proportion states that In a chemical substance the elements are always present in definite proportions by mass. This law is also known as 'Law of definite proportions '.
Mass of 1 atom of sulphur = 32 g
Mass of 1 atom of oxygen = 16 g
Mass of 2 atoms of oxygen =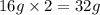
In formation of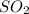 , 1 atom of sulfur combines with 2 atoms of oxygen and thus the mass ratio will be 32: 32= 1:1 .
, 1 atom of sulfur combines with 2 atoms of oxygen and thus the mass ratio will be 32: 32= 1:1 .
Thus the ratio of mass of sulfur atom to mass of oxygen atom in sulfur dioxide is 1: 1.