KindaSmartPersonn
23.05.2020 •
Chemistry
Question 5 (0 points)
What is the total pressure of gas in a propane tank if propane has a partial pressure of 22.4 atm, nitrogen's is 0.44 atm, and oxygen's is 0.19 atm and methane at a partial pressure of 2.86atm?
a. 22.26 atm
b. 18.91 torr
c. 28.33 atm
d. 25.89 atm
Solved
Show answers
More tips
- B Business and Finance How to Open an Online Store? A Detailed Guide for Beginners...
- W Work and Career How to Write a Resume That Catches the Employer s Attention?...
- C Computers and Internet Е-head: How it Simplifies Life for Users?...
- F Family and Home How to Choose the Best Diapers for Your Baby?...
- F Family and Home Parquet or laminate, which is better?...
- L Leisure and Entertainment How to Properly Wind Fishing Line onto a Reel?...
- L Leisure and Entertainment How to Make a Paper Boat in Simple Steps...
- T Travel and tourism Maldives Adventures: What is the Best Season to Visit the Luxurious Beaches?...
- H Health and Medicine Kinesiology: What is it and How Does it Work?...
- O Other How to Choose the Best Answer to Your Question on The Grand Question ?...
Answers on questions: Chemistry
- C Chemistry E decide whether these proposed lewis structures are reasonable. proposed lewis structure is the proposed lewis structure reasonable? h c n yes. no, it has the wrong number...
- C Chemistry Draw a Lewis structure for O3 and answer the following questions based on your drawing. 1. For the central oxygen atom: ... The number of lone pairs = The number of single...
- C Chemistry Write the balanced half-reaction equation for when H 2 O 2 ( aq ) acts as an oxidizing agent in an acidic solution. Phases are optional....
- C Chemistry What is true about 1.0 mol ca and 1.0 mol mg they are equal in mass they contain the same number of atoms they have the same atomic mass their molar masses are equal...
- C Chemistry I m very tired so if somebody could that would be greatly appreciated!...
- C Chemistry The reaction below is allowed to come to equilibrium. after equilibrium is reached, the temperature of the container is raised by 50ºc. which statement below describes a change...
- S Social Studies The way different groups work together in a society is known as its? a) institution b) civilization c) social class d) social structure...
- C Chemistry In collecting the precipitate, why would it be inappropriate to heat the reacted mixture and evaporate off the water?...
- M Mathematics What is the absolute value of -2* -2 *-2*-2...
- A Advanced Placement (AP) Earth spins on its axis, and one rotation takes about one...

Ответ:
37.44 grams of sodium chloride will be produced.
Explanation:The mole concept of the compound is necessary for solving these types of problems.
Atomic weight of sodium = 23.
Atomic weight of chlorine = 35.5.
So, molecular weight of chlorine = 71.
So number of moles of sodium present =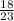
=0.78
So,number of moles of chlorine present =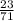
= 0.32
1 mole of chlorine reacts with 2 moles of sodium to form 2 moles of sodium chloride.
So, 0.32 moles of chlorine will react with 0.64 moles of sodium to form 0.64 moles of sodium chloride.
As sodium is present in excess, so chlorine is the limiting reagent.
Molecular weight of sodium chloride=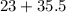 = 58.5.
= 58.5.
So, weight of sodium chloride produced = grams = 37.44 grams.
grams = 37.44 grams.