Kaitneedshelps
18.10.2019 •
Chemistry
Rearrange in to a quadratic 5.333 = (0.6 + 2p)² / (0.135 - p)
Solved
Show answers
More tips
- G Goods and services Kogda zhdatt Iphone 5? The Latest News and Rumors...
- F Family and Home Parquet or laminate, which is better?...
- L Leisure and Entertainment How to Properly Wind Fishing Line onto a Reel?...
- L Leisure and Entertainment How to Make a Paper Boat in Simple Steps...
- T Travel and tourism Maldives Adventures: What is the Best Season to Visit the Luxurious Beaches?...
- H Health and Medicine Kinesiology: What is it and How Does it Work?...
- O Other How to Choose the Best Answer to Your Question on The Grand Question ?...
- L Leisure and Entertainment History of International Women s Day: When Did the Celebration of March 8th Begin?...
- S Style and Beauty Intimate Haircut: The Reasons, Popularity, and Risks...
- A Art and Culture When Will Eurovision 2011 Take Place?...
Answers on questions: Chemistry
- C Chemistry What determines the change of an ion formed by an atom...
- P Physics I need to know if i have this correct...
- M Mathematics a fishing boat travels upstream running 20km/hr. the river was moving 8km/hr. what was the velocity of the boat...
- S Spanish Choose the word that best completes the phrase. el agricultor termina su (1st) año de estudios. primer octavo tercer primero...
- M Mathematics 2, 344 a whole number, rational number, or integer...

Ответ:
Ответ:
A. The Salicylic acid is the limiting reagent.
is the limiting reagent.
B. 4.66 g of the acetic anhydride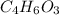 remain.
remain.
C. 2.6 g of are produced.
are produced.
Explanation:
A. First write the balanced chemical equation fior the asprin synthesis:
Then determine the limiting reagent.
To determine the limiting reagent first divide the mass of each compound between its molar mass, then divide this quantity by the number of moles given by the reaction and the smallest number will be the limiting reagent.
- For the Salicylic acid :
:
- For the Acetic anhydride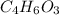 :
:
The smallest number is for the salicylic acid therefore it is the limiting reagent.
B. Calculate how many grams of the excess reagent remain.
- First calculate how many grams of acetic anhydride reacts:
- Then subtract the quantity of acetic anhydride that reacts from the quantity that are mixed:
C. Calculate how many grams of product are produced: