xelynncaldera
01.11.2019 •
Chemistry
Robert places a piece of magnesium metal (mg) into a test tube of hydrochloric acid (hcl). the magnesium begins to bubble and fizz, releasing hydrogen gas (h2) and the test tube gets warm. which equation represents the reaction robert observed?
Solved
Show answers
More tips
- F Food and Cooking Discover Delicious Recipes You Can Make with Ground Meat...
- C Computers and Internet Google Search Tips and Tricks: Everything You Need to Know...
- S Science and Technology Why is there no gravity on other planets?...
- L Leisure and Entertainment How to Properly Wind Fishing Line onto a Reel?...
- L Leisure and Entertainment How to Make a Paper Boat in Simple Steps...
- T Travel and tourism Maldives Adventures: What is the Best Season to Visit the Luxurious Beaches?...
- H Health and Medicine Kinesiology: What is it and How Does it Work?...
- O Other How to Choose the Best Answer to Your Question on The Grand Question ?...
- L Leisure and Entertainment History of International Women s Day: When Did the Celebration of March 8th Begin?...
- S Style and Beauty Intimate Haircut: The Reasons, Popularity, and Risks...
Answers on questions: Chemistry
- B Business Aless-established company, or a company with a high debt to equity ratio, would be considered a riskier investment to the lender. which of the following principles attests...
- P Physics The ___ of a position time graph represents an objects velocity...
- M Mathematics Find the 23 rd term of the arithmetic sequence whose common difference is d=5 and whose first term is a1=1...
- H History Using the map which city is Mark letter c? help help help help...

Ответ:
Explanation:
The reaction is a simple replacement reaction in which hydrogen gas is liberated when it gets replaced by Magnesium in aqueous solution of hydrochloric acid.
1 mole of Mg reacts with 2 moles of HCl to give 1 mole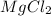 and 1 mole
and 1 mole  gas. The balanced equation for the reaction Robert observed is as follows:
gas. The balanced equation for the reaction Robert observed is as follows:
Ответ:
66
Explanation: