The Copper Chloride solution used in the investigation contained 300 grams per dm3 of solid CuCl2 dissolved in 1dm3 of water. The student used 50cm3 of copper chloride solution in each experiment. Calculate the mass of solid copper chloride used in each experiment.
Solved
Show answers
More tips
- F Family and Home How to Choose Suitable Windows for Your Home?...
- C Computers and Internet Why is Yandex s robot Bless.yandex.ru Important?...
- F Food and Cooking Fresh and Easy Lemonade in 10 Minutes...
- S Sport How to wrap boxing hand wraps? Everything you need to know!...
- F Family and Home How to Sew Curtain Tapes: Best Tips from Professionals...
- A Animals and plants How to Grow Lime from a Seed: Simple Tips and Interesting Facts...
- C Computers and Internet How to Create a Folder on Your iPhone?...
- G Goods and services How to sew a ribbon: Tips for beginners...
- F Food and Cooking How to Make Mayonnaise at Home? Secrets of Homemade Mayonnaise...
- C Computers and Internet Which Phone is Best for Internet Surfing?...
Answers on questions: Chemistry
- M Mathematics PLEASEEE HELP ME AND CAN YOU PLEASE SHOW YOUR WORK :)...
- S Social Studies The child of about 8 or 9 years of age begins to consider several tasks and relationships simultaneously. at this point in development children grasp not only their own social...
- B Biology What could potentially cause more damage (or a greater benefit) to an organism, a deletion mutation at the beginning of a DNA sequence or at the end of a DNA sequence?...
- M Mathematics 2(4-3x)=2(2x+5) as easy as this looks i keep getting the wrong answer : / i have -9 when it s supposed to be -1/5...

Ответ:
The mass of solid copper chloride used in each experiment=15 g
Explanation:
We are given that
Mass of copper chloride in solution=300g/dm3
Volume of solution used in each experiment=50 cm3
We have to find the mass of solid copper chloride used in each experiment.
1 dm3 contained solid copper chloride=300 g
0.05 dm3 contained solid copper chloride=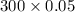 g
g
0.05 dm3 contained solid copper chloride=15 g
Hence, the mass of solid copper chloride used in each experiment=15 g
Ответ: