JimmySample7
25.03.2020 •
Chemistry
The elementary reaction 2 H 2 O ( g ) − ⇀ ↽ − 2 H 2 ( g ) + O 2 ( g ) proceeds at a certain temperature until the partial pressures of H 2 O , H 2 , and O 2 reach 0.025 atm, 0.0060 atm, and 0.0090 atm, respectively at equilibrium. What is the value of the equilibrium constant at this temperature?
Solved
Show answers
More tips
- F Food and Cooking Discover Delicious Recipes You Can Make with Ground Meat...
- C Computers and Internet Google Search Tips and Tricks: Everything You Need to Know...
- S Science and Technology Why is there no gravity on other planets?...
- L Leisure and Entertainment How to Properly Wind Fishing Line onto a Reel?...
- L Leisure and Entertainment How to Make a Paper Boat in Simple Steps...
- T Travel and tourism Maldives Adventures: What is the Best Season to Visit the Luxurious Beaches?...
- H Health and Medicine Kinesiology: What is it and How Does it Work?...
- O Other How to Choose the Best Answer to Your Question on The Grand Question ?...
- L Leisure and Entertainment History of International Women s Day: When Did the Celebration of March 8th Begin?...
- S Style and Beauty Intimate Haircut: The Reasons, Popularity, and Risks...
Answers on questions: Chemistry
- C Chemistry What is the molecular weight of benzoic acid...
- C Chemistry Which can not be chemically broken down into simpler substances...
- C Chemistry In order to change a solid to a liquid or a liquid to a gas, what is needed? A high boiling point An increase in thermal energy A high melting point A decrease in thermal energy...
- C Chemistry All energy travels in waves true or false...
- C Chemistry CAN YOU PLEASE HELP ME ON THIS I REALLY DONT WANT TO FAIL AND I HAVE TO GO WITH MY DAD AND THATS ALREADY BAD SO PLEASE HELP ME IN THIS I WILL GIVE YOU ALL MY POINT I PROMISE...
- C Chemistry What occurs at the cathode of a galvanic cell? (sorry about the picture my back camera is broken)...
- C Chemistry HELPPP PLS 7.) All isotopes of element T are radioactive, but they have widely varying half-lives. If an 1600.0 g sample of element T decays to 100.0 g of in 639,000 years,...
- C Chemistry An aquifer receives 40 m3 of precipitation and loses 10 m3 of water through natural movement. How much water can be pumped from the aquifer to balance the budget? 10 m3 20...
- M Mathematics Determine the relation for the function y= -3+4 using x-values of (-2,0,3,5)...
- M Mathematics Which value of x, from the choices below, make the expressions x + 3 and 2x equivalent?...

Ответ:
The value of equilibrium constant = 0.005184
Explanation:
According to the given reaction we consider Kp equation and find the value of equilibrium constant using partial pressure of individual gases.
2 H 2 O ( g ) ⇄ 2 H 2 ( g ) + O 2 ( g )
Kp =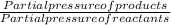
Change of gaseous moles(Δn)
= 3 - 2
= 1
⇒
∴ The value of equilibrium constant = 0.005184
Ответ: