The lowest pressure attainable using the best available vacuum techniques is about 10−12 n/m2. part a at such a pressure, how many molecules are there per cm3 at 2 ∘c?
Solved
Show answers
More tips
- S Style and Beauty How to choose the best mascara for your eyelashes...
- F Food and Cooking Discover Delicious Recipes You Can Make with Ground Meat...
- C Computers and Internet Google Search Tips and Tricks: Everything You Need to Know...
- S Science and Technology Why is there no gravity on other planets?...
- L Leisure and Entertainment How to Properly Wind Fishing Line onto a Reel?...
- L Leisure and Entertainment How to Make a Paper Boat in Simple Steps...
- T Travel and tourism Maldives Adventures: What is the Best Season to Visit the Luxurious Beaches?...
- H Health and Medicine Kinesiology: What is it and How Does it Work?...
- O Other How to Choose the Best Answer to Your Question on The Grand Question ?...
- L Leisure and Entertainment History of International Women s Day: When Did the Celebration of March 8th Begin?...
Answers on questions: Chemistry
- C Chemistry Based on the information provided, which solution is a base and a weak electrolyte?...
- C Chemistry How many grams of nano3 would have to be added to 100. grams of water at 45°c to make a saturated solution of this salt? 1. 100. 2. 110. 3. 120. 4. 130....
- C Chemistry Help these students choose the correct type of graph to use to depict their data sets. Darla wants to show what percentage of her mouse population has white, brown, black,...
- C Chemistry 2C2H6: How many atoms for each element are in the formula? C= H=...
- C Chemistry Answer the following- what is the dot structure, cross-cross method, molecular mass, compound name, compound formulae for each reactants: potassium and oxygen, sodium and...
- C Chemistry Kiera, a 330 N girl, steps in water that someone spilt on the floor. The coefficient of kinetic friction between Kiera and the floor is 0.028. What is the force of kinetic...
- C Chemistry The temperature of a gas inside of a balloon is increased from 250K to 298K. If the initial volume of the balloon was 1.75 Liters, what is the volume of the balloon after...
- H History How does the use of perspective affect the appearance of individuals in Renaissance paintings? They appear to be flat. They appear to be larger. They appear to be closer...
- C Computers and Technology What types of input and output devices would be ideal for a college student completing his or her coursework?...
- M Mathematics The measure of two angles are (6x-12) and (2x+32)°. What is the value of x if the two angles are supplementary?...

Ответ:
Number of molecules: N = 263.31
Further explanationSome of the laws regarding gas can apply to ideal gas (volume expansion does not occur when the gas is heated),:
Boyle's law at constant T,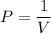
Charles's law, at constant P,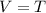
Avogadro's law, at constant P and T,So that the three laws can be combined into a single gas equation, the ideal gas equation
In general, the gas equation can be written
where
P = pressure, atm , N/m²
V = volume, liter
n = number of moles
R = gas constant = 0.082 l.atm / mol K (P= atm, v= liter),or 8,314 J/mol K (P=Pa or N/m2, v= m³)
T = temperature, Kelvin
n = N / No
n = mole
No = Avogadro number (6.02.10²³)
n = m / m
m = mass
M = relative molecular mass
Known
P = 10−12 N / m2
V = 1 cm3 = 10-6 m3
T = 2 ºC = 2 + 273 = 275 K
R = 8,314 J / mol. K
then the number of molecules (N):
N = n x No
N = 4,374.10⁻²² x 6.02.10²³
N = 263.31
Learn moreWhich equation agrees with the ideal gas law
link
link
Which law relates to the ideal gas law
link
Ответ:
25 mL of 1 × 10–5 M Co(NO₃)₂ and 75 mL of 5 × 10–4 M Na₂S
500 mL of 7.5 × 10–4 M AlCl₃ and 100 mL of 1.7 × 10–5 M Hg₂(NO₃)₂.
650 mL of 0.0080 M K₂SO₄ and 175 mL of 0.15 M AgNO₃
Explanation:
When 2 compounds that produce an insoluble substance are mixed together, A precipitate will be formed if Q of reaction > Ksp
For the solutions:
1.5 L of 0.025 M BaCl₂ and 1.25L of 0.014 M Pb(NO₃)₂.
Ksp is:
PbCl₂(s) ⇄ Pb²⁺(aq) + 2Cl⁻(aq)
Ksp = 2.4x10⁻⁴ = [Pb²⁺][Cl⁻]²
Molar concentration of each ion is:
[Pb²⁺] = 1.25L ₓ (0.014mol / L) = 0.0175mol / 2.75L = 6.36x10⁻³M
[Cl⁻] = 2 ₓ 1.5L ₓ (0.025mol / L) = 0.075mol / 2.75L = 0.0273M
Replacing in Ksp expression to find Q:
Q = [6.36x10⁻³M][0.0273M]² = 4.73x10⁻⁶
As Q < Ksp, the mixture will not produce a precipitate.
25 mL of 1 × 10–5 M Co(NO₃)₂ and 75 mL of 5 × 10–4 M Na₂S
Ksp is:
CoS(s) ⇄ Co²⁺(aq) + S²⁻(aq)
Ksp = 4.0x10⁻²¹ = [Co²⁺][S²⁻]
Molar concentration of each ion is:
[Co²⁺] = 0.025L ₓ (1x10⁻⁵mol / L) = 2.5x10⁻⁷mol / 0.1L = 2.5x10⁻⁶M
[S²⁻] = 0.075L ₓ (5x10⁻⁴mol / L) = 3.75x10⁻⁵mol / 0.1L = 3.75x10⁻⁴M
Replacing in Ksp expression to find Q:
Q = [2.5x10⁻⁶M][3.75x10⁻⁴M] = 9.38x10⁻⁶
As Q > Ksp, the mixture will produce a precipitate.
500 mL of 7.5 × 10–4 M AlCl₃ and 100 mL of 1.7 × 10–5 M Hg₂(NO₃)₂.
Ksp is:
Hg₂Cl₂(s) ⇄ 2Hg⁺(aq) + 2Cl⁻(aq)
Ksp = 3.5x10⁻¹⁸ = [Hg⁺]²[Cl⁻]²
Molar concentration of each ion is:
[Hg⁺] = 2ₓ0.100L ₓ (1.7x10⁻⁵mol / L) = 3.4x10⁻⁶mol / 0.6L = 5.67x10⁻⁶M
[Cl⁻] = 3 ₓ 0.500L ₓ (7.5x10⁻⁴mol / L) = 1.125x10⁻³mol / 0.6L = 1.88x10⁻³M
Replacing in Ksp expression to find Q:
Q = [5.67x10⁻⁶M]²[1.88x10⁻³M]² = 1.14x10⁻⁶
As Q > Ksp, the reaction will produce a precipitate.
650 mL of 0.0080 M K₂SO₄ and 175 mL of 0.15 M AgNO₃
Ksp is:
Ag₂SO₄(s) ⇄ 2Ag⁺(aq) + SO₄²⁻(aq)
Ksp = 1.5x10⁻⁵ = [Ag⁺]²[SO₄²⁻]
Molar concentration of each ion is:
[Ag⁺] = 0.175L ₓ (0.15mol / L) = 0.02625mol / 0.825L = 0.0318M
[SO₄²⁻] = 0.650L ₓ (0.080mol / L) = 0.052mol / 0.825L = 0.0630M
Replacing in Ksp expression to find Q:
Q = [0.0318M]²[0.0630M] = 6.37x10⁻⁵
As Q > Ksp, the reaction will produce a precipitate.