The type of hybridisation and bond angle in [Ni(CN)4]−2 are
Solved
Show answers
More tips
- F Food and Cooking Apple Cider Vinegar: The Ultimate Health and Beauty Solution...
- C Computers and Internet Е-head: How it Simplifies Life for Users?...
- F Family and Home How to Choose the Best Diapers for Your Baby?...
- F Family and Home Parquet or laminate, which is better?...
- L Leisure and Entertainment How to Properly Wind Fishing Line onto a Reel?...
- L Leisure and Entertainment How to Make a Paper Boat in Simple Steps...
- T Travel and tourism Maldives Adventures: What is the Best Season to Visit the Luxurious Beaches?...
- H Health and Medicine Kinesiology: What is it and How Does it Work?...
- O Other How to Choose the Best Answer to Your Question on The Grand Question ?...
- L Leisure and Entertainment History of International Women s Day: When Did the Celebration of March 8th Begin?...
Answers on questions: Chemistry
- C Chemistry Define physical quantity?...
- C Chemistry Which have the lowest first ionization energies ? 1.Alkaline Earth metals 2.Transition metals 3.Halogens 4.Alkali metals 5.Noble gases...
- C Chemistry If 2.5 grams of Cu(NO3)2 are reacted, how many grams of NaNO3 would be formed? Show your work....
- C Chemistry Electronegativity is A) a value of an element that indicates if it is a metal or a nonmetal. B) a value of an element that indicates if it is a metalloid. C) a value of an element...
- C Chemistry Three physiacal changes that are in nature...
- C Chemistry What group is more reactive than the alkaline earth metals...
- C Chemistry Moritz is watching his little brother play in a mud puddle. moritz notices that when his brother stirs it with a stick, after a few moments the dirt settles back to the bottom of...
- C Chemistry Identify the limiting reactant for the following: 0.308 moles Na3PO4 mix with 0.587 moles FeCl2 2 Na3PO4 + 3 FeCl2 → Fe3(PO4)2 + 6 NaCl...
- C Chemistry Convert 2.3*10^9 seconds into years....
- C Chemistry PLZZZZZ HELPPP ILL GIVE BRAINLIEST JUST TELL ME IF THIS IS CORRECT BECAUSE IF I GET IT WRONG ILL HAVE A HORRIBLE GRADE PLZ...

Ответ:
The average atomic mass of M is 181.33 g/mol.
Explanation:
First off we need to know the reaction that takes place. The balanced reaction of M₂S₃(s) is:
M₂S₃(s) + O₂(g) ⇒2MO₂(s) + 3SO₂(g)The important section for this problem is this:
M₂S₃(s) ⇒2MO₂(s)**Thus, the number of moles of M in M₂S₃ is equal to the number of moles of M in 2MO₂.
The decrease in mass means that M₂S₃ reacted and produced MO₂, thus the mass of MO₂ is 3.280-0.228=3.052 g
Now let's say x is the atomic weight of M, and write the molecular weights (Mw) of those two compounds:
Mw of M₂S₃= 2x + 96 g/mol
Mw of MO₂= x + 32 g/mol
Now we determine the moles of each compound, using the formula [ moles = mass / molecular weight ]:
Moles of M₂S₃=
Moles of MO₂=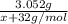
Using the equivalence marked by asterisks, we're left with (note that the second denominator is multiplied by 2 because of the reaction coefficients):
We solve for x:
Thus, the average atomic mass of M is 181.33 g/mol.