Two humid streams are adiabatically mixed at 1 atm pressure to form a third stream. The first stream has a temperature of 40C, a relative humidity of 40%, and a volumetric flow rate of 3 L/s, while the second stream has a temperature of 15C, a relative humidity of 80%, and a volumetric flow rate of 1 L/s. Calculate the third stream’s temperature and relative humidity
Solved
Show answers
More tips
- S Style and Beauty How to choose the best mascara for your eyelashes...
- F Food and Cooking Discover Delicious Recipes You Can Make with Ground Meat...
- C Computers and Internet Google Search Tips and Tricks: Everything You Need to Know...
- S Science and Technology Why is there no gravity on other planets?...
- L Leisure and Entertainment How to Properly Wind Fishing Line onto a Reel?...
- L Leisure and Entertainment How to Make a Paper Boat in Simple Steps...
- T Travel and tourism Maldives Adventures: What is the Best Season to Visit the Luxurious Beaches?...
- H Health and Medicine Kinesiology: What is it and How Does it Work?...
- O Other How to Choose the Best Answer to Your Question on The Grand Question ?...
- L Leisure and Entertainment History of International Women s Day: When Did the Celebration of March 8th Begin?...
Answers on questions: Chemistry
- C Chemistry Acompound a has the formula c8h10. after vigorous oxidation, it yields phthalic acid. draw the structure of a....
- C Chemistry What happened to the glycerol formed during the saponification reaction?...
- C Chemistry What does the type of bond have to do with the properties of the compound?...
- C Chemistry In solid NaCl, each sodium ion is surrounded by six chlorine ions and each chlorine ion is surrounded by six sodium ions. This arrangement results in a Question 24 options:...
- C Chemistry 2. The carbon cycle is important to an ecosystem because but SO...
- C Chemistry The ion of an element has 3 positive charges. Mass number of the atom is 27 and the number of neutrons is 14. What is the number of electrons in the ion?....
- C Chemistry Diamond has a density of 3.26 g/cm^3. what is the mass of a diamond that has a volume of 0.351 cm^3?...
- C Chemistry What happens to a glass of water as the average molecular kinetic energy of the water molecules increases? the water becomes more acidic. the water becomes warmer. the...
- E English Of mice and men graphic organizer...
- M Mathematics Suppose another hiker takes 92 minutes to complete the hike .find the mean and media times including this new time...

Ответ:
-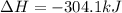
Solution:- The enthalpy changed asked to calculate for the reaction,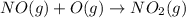
It is based on Hess's law where we use the given equations to make the desired equation and calculate it's enthalpy change.
For our equation we need one NO(g) on reactant side and it is present in first equation so let's write this as such.
Now we need O(g) on reactant side which is present in third equation. Third equation has two O on product side so we need to divide it by 2 and also reverse it so that we could get one O on reactant side. When an equation is reversed then sign of delta H also gets changed.
To cancel ozone, we need to reverse second equation.
Let's add all these three equations now:
------------------------------------------------------------------------------------------------------------
So, the enthalpy change for the given equation asked to calculate is -304.1 kJ.