cjmckee2001
23.09.2019 •
Chemistry
What is the electron configuration of the element located in the fourth row of the periodic table in group 5a?
Solved
Show answers
More tips
- F Food and Cooking Fresh and Easy Lemonade in 10 Minutes...
- S Sport How to wrap boxing hand wraps? Everything you need to know!...
- F Family and Home How to Sew Curtain Tapes: Best Tips from Professionals...
- A Animals and plants How to Grow Lime from a Seed: Simple Tips and Interesting Facts...
- C Computers and Internet How to Create a Folder on Your iPhone?...
- G Goods and services How to sew a ribbon: Tips for beginners...
- F Food and Cooking How to Make Mayonnaise at Home? Secrets of Homemade Mayonnaise...
- C Computers and Internet Which Phone is Best for Internet Surfing?...
- F Food and Cooking Everything You Need to Know About Pasta...
- C Computers and Internet How to Choose a Monitor?...
Answers on questions: Chemistry
- P Physics 2D Motion - Comparing the path of projectiles on Earth and Mars Imagine you are repeating the Land the Rover experiment on Mars. How would the experiment differ than the one done on...
- M Mathematics The equation of the graphed line is 2x-3y=12 what is the x-intercept of the graph ?...
- H Health What does media s ideal body image look like? Own words please?...
- M Mathematics Someone do this got me rq...
- S SAT Janet was diagnosed with breast cancer nine months ago. Now the cancer has spread to her lymph glands as well. What is this process called when cancer spreads to other (secondary)...

Ответ:
The electronic configuration of the element located in the fourth row of group 5A is![\boxed{\left[ {{\text{Ar}}}\right]3{d^{10}}4{s^2}4{p^3}}](/tpl/images/0255/0660/a4888.png) .
.
Further explanation:
The electronic configuration is the distribution of electrons of an atom in the atomic orbitals. There are two states for an electron: ground as well as the excited state. The configuration of the atom in the lowest possible energy levels is called the ground-state electronic configuration. When an electron jumps from the stable ground state to some higher level, that state is called the excited state, and the electronic configuration corresponding to this state is known as the excited-state half-filled configuration.
The filling of electrons in different energy levels or orbitals is done in accordance with the following three rules.
1. Aufbau principle: The principle states that the electrons are filled in various orbitals in the increasing order of their energies as follows:
2. Hund’s rule: Electron pairing will not start until each orbital is singly occupied.
3. Pauli’s exclusion principle: According to this principle, all the four quantum numbers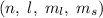 for any two electrons can never be the same. In an orbital, the spin of two electrons has to be different. If one electron has the clockwise spin, the other would have the anticlockwise spin and vice-versa.
for any two electrons can never be the same. In an orbital, the spin of two electrons has to be different. If one electron has the clockwise spin, the other would have the anticlockwise spin and vice-versa.
Group 5A belongs to the pnictogen family. It includes elements like nitrogen (N), phosphorus (P), arsenic (As), antimony (Sb), bismuth (Bi). The general electronic configuration of this group is . The element that is present in the fourth row of this group is arsenic (As), and its electronic configuration is
arsenic (As), and its electronic configuration is ![\left[{{\text{Ar}}}\right]3{d^{10}}4{s^2}4{p^3}](/tpl/images/0255/0660/aada3.png) .
.
Learn more:
1. Determine the ion with configuration![\left[{{\text{Ar}}}\right]\;3{d^2}](/tpl/images/0255/0660/89724.png) :
:
2. Identification of element which has electron configuration :
:
Answer details:
Grade: High School
Subject: Chemistry
Chapter: Electronic configuration of the elements
Keywords: 5A, arsenic, pnictogen, nitrogen, phosphorus, arsenic, antimony, bismuth, N, P, As, Sb, Bi, ns2np3, aufbau principle, Hund’s rule, Pauli’s exclusion principle, electronic configuration, distribution of electrons.
Ответ:
I have actually :) he is indeed bae whats your favourite song?